
SubQ It! Subcuticular Surgical Stapler - SU-10
- FDA 510(k) cleared, ISO 13485 certified
- Deploys in 7 sec vs 42 sec per stitch
- 10 bioabsorbable fasteners, up to 10 cm
FDA 510(k) Cleared
ISO 13485 Certified
SubQ It! bioabsorbable skin staplers by Opus KSD, Inc. are FDA-cleared disposable surgical devices pre-loaded with bioabsorbable dermal fasteners for subcuticular incision closure. Available in SU-10 and SU-25 models, they close incisions up to 25 cm across abdominal, gynecologic, orthopedic, and plastic surgery applications — 7X faster than manual sutures with no staple removal required.




A bioabsorbable skin stapler deploys small polymer fasteners subcutaneously — beneath the skin surface — to close surgical incisions without piercing external skin. The fasteners hold tissue together during healing and are then naturally absorbed by the body, eliminating the need for staple removal and reducing the risk of scarring or infection associated with percutaneous metal staples.
Talk to our experts for custom solutions and tailored guidance.

Rapid subcuticular closure of trocar incisions, laparoscopic cholecystectomy, and abdominal surgery sites.
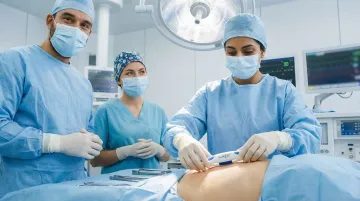
Cosmetically superior closure for Cesarean sections and gynecologic procedures without metal staple removal.
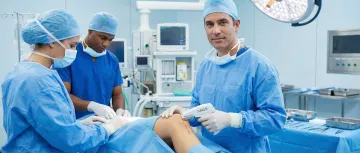
Fast, scarring-minimized skin closure for orthopedic incisions, eliminating the need for percutaneous staple removal.
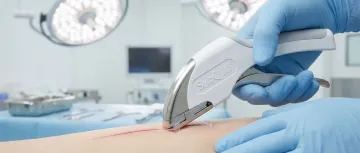
Subcuticular placement ensures no external skin piercing, delivering superior cosmetic outcomes in reconstructive procedures.
Talk to our experts for custom solutions and tailored guidance.
Each bioabsorbable fastener deploys in just 7 seconds, dramatically reducing OR closure time and lowering overall surgical costs.
Bioabsorbable fasteners are absorbed by the body after healing, eliminating return visits, patient discomfort, and removal procedure costs.
Cleared for abdominal, thoracic, gynecologic, orthopedic, and plastic surgery. Manufactured under ISO 13485-registered quality standards.
Submit your inquiry and our sales team will respond with pricing, IFU documentation, and training resources within one business day.
For immediate assistance, feel free to give us a direct call at +1 339.933.8811. You can also send us a quick email at rob@subq-it.com.
For immediate assistance, feel free to give us a direct call at +1 339.933.8811. You can also send us a quick email at rob@subq-it.com.