
SubQ It! Subcuticular Surgical Stapler - SU-10
- 10 bioabsorbable fasteners, no follow-up visit
- Deploys in 7 sec, 7X faster than sutures
- Retractable needles with multi-safety mechanism
ISO 13485 Certified
FDA 510(k) Cleared
The SubQ It! bioabsorbable subcuticular surgical stapler delivers rapid, scarring-minimized incision closure without the need for staple removal. Available in SU-10 and SU-25 configurations, these FDA-cleared, ISO 13485-certified devices deploy bioabsorbable fasteners subcutaneously — ideal for minimally invasive, abdominal, gynecologic, orthopedic, and reconstructive procedures.




Staples and sutures heal at a comparable rate, but closure speed differs significantly. The SubQ It! bioabsorbable stapler deploys each fastener in 7 seconds — 7X faster than manual sutures — reducing OR time. Because fasteners are placed subcuticularly, they avoid skin-surface trauma, which supports cleaner healing and minimized scarring compared to traditional metal staples.
Talk to our experts for custom solutions and tailored guidance.
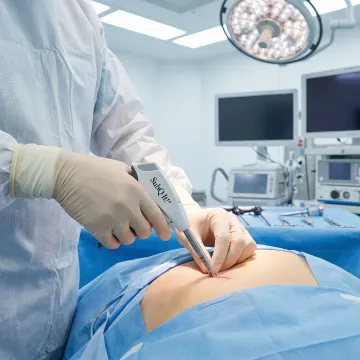
Closes trocar and laparoscopic incisions subcuticularly in seconds, reducing MIS procedure time and patient recovery.
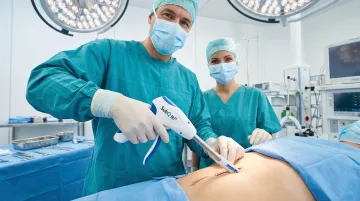
Efficiently closes longer abdominal and thoracic incisions with bioabsorbable fasteners for improved cosmetic outcomes.
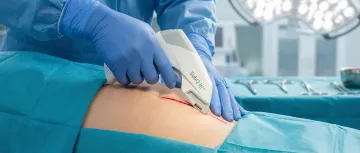
Supports Cesarean sections and gynecologic procedures with fast, scarring-minimized subcuticular skin closure.
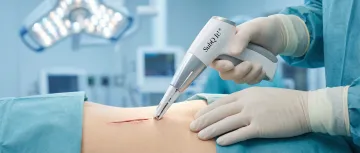
Delivers cosmetically superior, scar-minimizing closures for reconstructive and plastic surgical incisions of varying lengths.
Talk to our experts for custom solutions and tailored guidance.
Each bioabsorbable fastener deploys in just 7 seconds, dramatically cutting closure time and reducing overall OR costs compared to traditional suturing methods.
Bioabsorbable fasteners are fully absorbed by the body after healing, eliminating follow-up removal visits, patient discomfort, and all associated staple-removal costs.
SubQ It! holds FDA 510(k) clearance (K131563) and is manufactured under ISO 13485-certified quality systems, ensuring regulatory compliance and device reliability.
Submit your details and our sales team will respond with pricing, IFU documentation, and in-service training options tailored to your facility.
For immediate assistance, feel free to give us a direct call at +1 339.933.8811. You can also send us a quick email at rob@subq-it.com.
For immediate assistance, feel free to give us a direct call at +1 339.933.8811. You can also send us a quick email at rob@subq-it.com.