
A search for a skin stapler on Amazon returns dozens of disposable metal devices. However, most of them are marketed for first aid, veterinary care, and general medical use. Additionally, surface-applied metal staples carry well-documented trade-offs.
According to studies, wound infections hit 3.46% in stapled cases compared to just 2.06% in sutured cases across a 2025 vascular surgery study of more than 18,000 procedures. The choice of a stapler affects more than the cost per unit.
This post breaks down what Amazon actually sells, how those products compare to surgical-grade devices, and when bioabsorbable subcutaneous closure fits for surgeons who want faster closure with fewer follow-up visits.
Key Takeaways
- Amazon skin staplers are disposable, surface-applied metal devices, designed for first aid, veterinary, and general medical use, not for bioabsorbable or subcutaneous closure.
- Stapled wounds developed infections at 3.46% vs. 2.06% for sutured wounds in a 2025 vascular study of 18,268 cases, with staples also tied to higher return-to-OR and longer hospitalization rates.
- Bioabsorbable subcutaneous staplers place fasteners under the skin that dissolve naturally, so there's no removal visit and no surface puncture scarring.
- Skin closure dropped from 36.5 minutes (hand-sewn suture) to 16.2 minutes (bioabsorbable subcutaneous stapler) across 4,311 mastectomy patients, with no meaningful difference in infection rates between groups.
- Surgeons evaluating skin staplers should compare the total cost of closure (device cost, OR time, removal visits, scarring outcomes) rather than unit price alone.
What Is a Skin Stapler?
A skin stapler is a handheld device that fires metal fasteners through the outer edges of a wound to hold the skin together while it heals. It's one of the fastest ways to close an incision. In the OR, skin staplers have been a standard surgical skin stapler option for decades.
The mechanism is straightforward. The surgeon positions the device over the wound edges, squeezes the handle, and a metal staple bends into a rectangular shape as it penetrates both sides of the skin. The staple holds the edges in apposition while the tissue underneath heals.
Traditional metal staples are surface-applied. They sit on top of the skin, visible and palpable, until a clinician removes them with a staple remover at a follow-up appointment, typically 7 to 10 days after surgery. That removal step is built into the workflow for every patient closed with conventional staples.
Why Are Skin Staplers Sold on Amazon Different From Surgical-Grade Devices?
Amazon's skin stapler listings are overwhelmingly disposable, single-use metal devices packaged for first aid, veterinary, and general medical applications. They generally range from $15 to $90 and come preloaded with 35 to 55 wide-format staples. Some include a staple remover in the kit.
Here's how they compare to professional surgical closure systems.
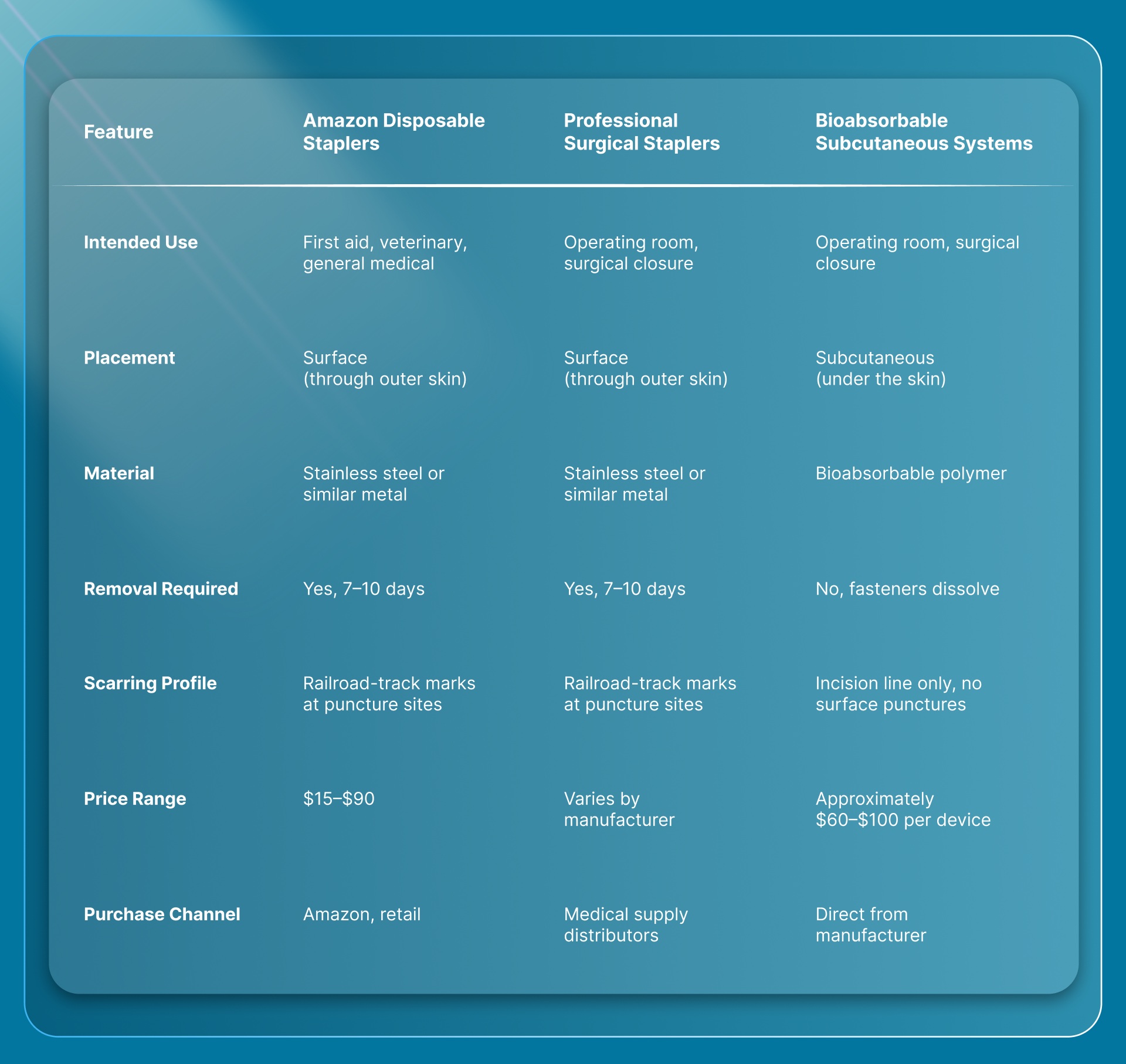
The key distinction goes beyond price. Amazon disposable skin staplers and professional OR staplers share the same basic design: metal fasteners on the skin surface, a mandatory removal visit, and puncture marks at every staple site. Bioabsorbable subcutaneous systems work on a different principle entirely, with fasteners placed under the skin that dissolve without a removal visit.
For surgeons who close incisions in the OR every week, the real comparison is between surface-applied closure (metal staples or sutures) and subcutaneous closure (bioabsorbable fasteners). Amazon only stocks the first category.
What Are the Risks of Using Traditional Metal Skin Staples?
Metal staples close wounds quickly, but the clinical data shows meaningful trade-offs in infection, reoperation, and scarring.
Infection and Reoperation
When researchers examined 18,268 lower extremity bypass cases across a 2025 multi-center vascular study, 3.46% of stapled wounds developed infections. Sutured wounds came in at 2.06%. Stapled patients also returned to the OR more often (15.17% vs. 11.53%) and stayed in the hospital longer than 7 days at a higher rate (28.72% vs. 22.51%).
Scarring
Every surface staple creates two puncture wounds, one on each side of the incision. Those puncture sites heal into characteristic railroad-track marks that remain visible long after the staple is gone. For plastic and cosmetic surgeons, that's a predictable cosmetic compromise. Sutures can produce similar marks, but subcuticular closure techniques minimize surface punctures.
Removal Burden
Metal staples require a follow-up visit for removal at 7 to 10 days. In a 2024 orthopedic randomized trial, surgeons finished staple closure in 5.2 minutes and suture closure in 12.6 minutes. The speed advantage was clear, but every patient in each group still came back on day 12 for removal.
The removal visit adds scheduling burden to the practice, discomfort for the patient, and cost to the episode of care. For high-volume practices, the cumulative time spent on staple removal adds up across the case load.
How Do Bioabsorbable Subcutaneous Staplers Compare to Surface Metal Staples?
Bioabsorbable subcutaneous staplers place fasteners beneath the skin's surface, into the dermal layer, where they hold tissue together during healing and then dissolve on their own. There's no surface puncture, no railroad-track scarring, and no removal visit.

SubQ It! is a bioabsorbable skin closure system that works on this principle. The SubQ It! SU-10 carries 10 fasteners for incisions up to 10 cm, including very small laparoscopic incisions where surface-applied devices can't operate. The SubQ It! SU-25 carries 25 fasteners and handles incisions up to 25 cm, covering open surgical procedures, plastic surgery, and longer closures.
Across 4,311 mastectomy patients in a study, surgeons using a bioabsorbable subcutaneous stapler wrapped up skin closure in 16.2 minutes on average. Hand-sewn suture took 36.5 minutes. Infections barely differed: 0.38% in the stapler group, 0.36% in the suture group.
Columbia University plastic surgeons found the same time advantage in a different procedure. In a 2024 cohort of 62 bilateral breast reduction patients, adding bioabsorbable dermal staples cut 21.8 minutes from total operative time vs. sutures alone. Even after controlling for tissue volume and pedicle type, the time savings held at 26.5 minutes. Complication rates didn't change.
Cesarean delivery data reinforces the trend. A 2025 cohort of 31,419 cases at a single tertiary center showed total surgery time averaging 52 minutes when surgeons used absorbable subcutaneous staples, 53 minutes with non-absorbable staples, and 60 minutes with sutures. Wound infection rates within 45 days were comparable across all three groups, and the bioabsorbable group had fewer prolonged hospitalizations and readmissions.
Across vascular, orthopedic, breast, and cesarean cases, the takeaway is the same. Bioabsorbable subcutaneous closure matches or beats surface staples and sutures on time, doesn't increase infection risk, and removes the follow-up visit from the equation entirely.
What Should Surgeons Look for When Choosing a Skin Closure Device?
The right closure device depends on how it performs across the full episode of care. Here are the criteria that matter.
Closure Speed and OR Time
Every minute of OR time carries a cost. The orthopedic and mastectomy data cited earlier show that the closure method alone can account for a double-digit minute difference per case. Bioabsorbable subcutaneous closure consistently finishes faster than hand-sewn suture, and staplers vs. sutures is no longer a binary choice. Faster closure frees up OR capacity for the next case.
Cosmetic Outcome
Surface staples leave paired puncture marks. Subcuticular sutures reduce that, but they take longer to place. Bioabsorbable subcutaneous fasteners avoid surface punctures entirely, leaving only the incision line itself. For plastic surgeons and any case where the patient cares about scar appearance, this matters.
Patient Follow-Up Burden
Metal staples and non-absorbable sutures both require a removal visit. Bioabsorbable fasteners dissolve on their own. Removing the follow-up visit from the care pathway saves scheduling time, reduces patient travel, and lowers the chance of a missed or delayed removal.
Incision Size Compatibility
Not every closure method works on every incision. Adhesive strips and tissue glues have length and tension limits. Some devices can't close very small laparoscopic incisions effectively.
Total Cost of Closure
A $15 Amazon stapler looks cheap until you factor in the removal visit, the reoperation risk, and the OR time difference. Every minute of OR time costs roughly $37.45 based on published surgical cost data. Saving 20 minutes on closure adds up fast, and that freed-up time can go toward additional cases.
Surgeons who evaluate closure devices on total cost rather than sticker price consistently find that faster, bioabsorbable options pay for themselves quickly.
Final Thoughts
Amazon's skin stapler listings reflect one slice of the closure market. They're disposable metal devices that fire surface staples, require removal in 7 to 10 days, and leave puncture-site scars. For surgeons who close incisions regularly, those trade-offs add up across patients, schedules, and cosmetic outcomes.
Before reordering from Amazon, consider whether surface-applied closure is the right method at all. Published data from vascular, orthopedic, breast, and cesarean surgery consistently show the same thing. Bioabsorbable subcutaneous closure matches or beats conventional options on speed, infection rates, and cosmetic results, while removing the follow-up visit entirely.
SubQ It! offers bioabsorbable subcutaneous closure in two sizes (SU-10 for incisions up to 10 cm, SU-25 for incisions up to 25 cm) that cover everything from small laparoscopic trocar sites to open surgical procedures. Every week a surgeon stays with surface staples is another week of scheduled removal visits and avoidable scarring. Contact us today to request a custom quote.
FAQs
1. Are Skin Staplers on Amazon Sterile and Safe for General Use?
Yes, most Amazon skin staplers come individually packaged and labeled as sterile for single use. They're designed for first-aid and general medical wound closure, not specifically for OR-grade surgical procedures requiring bioabsorbable or subcutaneous placement.
2. Do Bioabsorbable Fasteners Work on All Skin Types?
Yes, bioabsorbable subcutaneous fasteners are placed into the dermal layer beneath the skin surface, so skin type and pigmentation don't affect placement or function. The fasteners hold tissue together during healing and dissolve over time without removal.
3. How Long Does It Take for Subcutaneous Fasteners to Dissolve?
Bioabsorbable fasteners are designed to maintain wound support during the critical healing period and then break down gradually through the body's natural absorption process. The dissolution timeline depends on the polymer composition and the patient's tissue response.
4. Can a Skin Stapler Be Used on Pediatric Patients?
Yes, skin staplers can be used in pediatric cases where wound closure requires quick application and secure approximation. The choice between surface staples, sutures, or subcutaneous options depends on incision location, cosmetic considerations, and the patient's ability to tolerate a removal visit.
5. Do Subcutaneous Fasteners Require Any Special Storage Conditions?
No, subcutaneous bioabsorbable fasteners are stored in standard sealed sterile packaging at room temperature, similar to other single-use surgical devices. They don't require refrigeration, freezing, or any special handling beyond standard sterile technique during application.
6. Is Training Required to Use a Bioabsorbable Skin Closure System?
Yes, surgeons benefit from a brief orientation on the firing mechanism and subcutaneous placement technique, which differs from surface stapler application. Manufacturers typically offer training materials and support to ensure correct use during the first few cases.


