The final step of skin closure often carries broader implications, affecting operative efficiency, consistency across cases, and recovery management. The chosen method determines not only how efficiently the incision is closed, but also the steps that follow.
In many settings, surgical skin staplers are selected for their speed and predictable application. Their growing adoption reflects this demand, with the U.S. surgical staplers market projected to reach USD 4 billion by 2034.
At the same time, their use introduces considerations such as removal, surface penetration, and cosmetic outcomes.
In this blog, you’ll explore how to evaluate surgical skin stapler options in 2026, including device types, clinical trade-offs, and workflow considerations.
Key Takeaways:
- Surgical skin staplers enable faster, more consistent closure compared to sutures, especially in longer or high-volume procedures.
- They are commonly used for clean, linear incisions where rapid approximation is needed, but may not suit complex or high-precision closures.
- Traditional metal staples penetrate the skin, remain visible, and require removal within 7-14 days, adding a follow-up step.
- Staple removal extends the closure process beyond the operating room, increasing coordination, workload, and reliance on patient follow-through.
- Choosing the right closure method depends on balancing speed, control, tissue characteristics, and the overall impact on workflow beyond the procedure.
What Is a Surgical Skin Stapler?
A surgical skin stapler is a handheld device used in the operating room to close skin incisions quickly at the end of a procedure. Instead of sutures, it places metal staples across the wound to approximate skin edges, enabling faster closure.
The device drives each staple through the skin, where it is formed into a B-shaped configuration beneath the surface to secure the wound edges.
The staple bridge remains visible externally, while the legs anchor within the dermal layers. These staples require removal once sufficient healing has occurred.
Key characteristics of a surgical skin stapler include:

Once you understand what a surgical skin stapler is, it becomes easier to see how it functions during incision closure.
How a Surgical Skin Stapler Works During Incision Closure?
A surgical skin stapler is used at the end of a procedure to quickly and predictably close the skin layer. It replaces repeated needle passes with a controlled, mechanical deployment, helping standardize closure.
Here's how a surgical skin stapler works during incision closure:
- Wound edge alignment: The skin edges are aligned and slightly everted so both sides meet at the same level before firing the device.
- Device positioning: The stapler is placed perpendicular to the incision, with the jaws positioned evenly across both wound edges.
- Staple deployment: When the handle is compressed, a metal staple is driven through the epidermis and dermis on both sides of the incision.
- Staple formation: The staple legs bend underneath the skin into a rectangular configuration, securing the tissue and holding the edges together.
- Sequential closure: The device is moved along the incision, placing staples at regular intervals to complete closure.
This approach enables rapid, evenly spaced wound approximation. Because staples penetrate the skin, they remain visible and must be removed as healing progresses.
In contrast, systems such as SubQ It! SU-25 uses bioabsorbable fasteners placed beneath the skin, maintaining closure without external staples and eliminating the need for removal, while still supporting efficient deployment.
A clear view of how the device functions during incision closure also helps explain the different types used in clinical practice.
4 Key Types of Surgical Skin Staplers Used in Practice
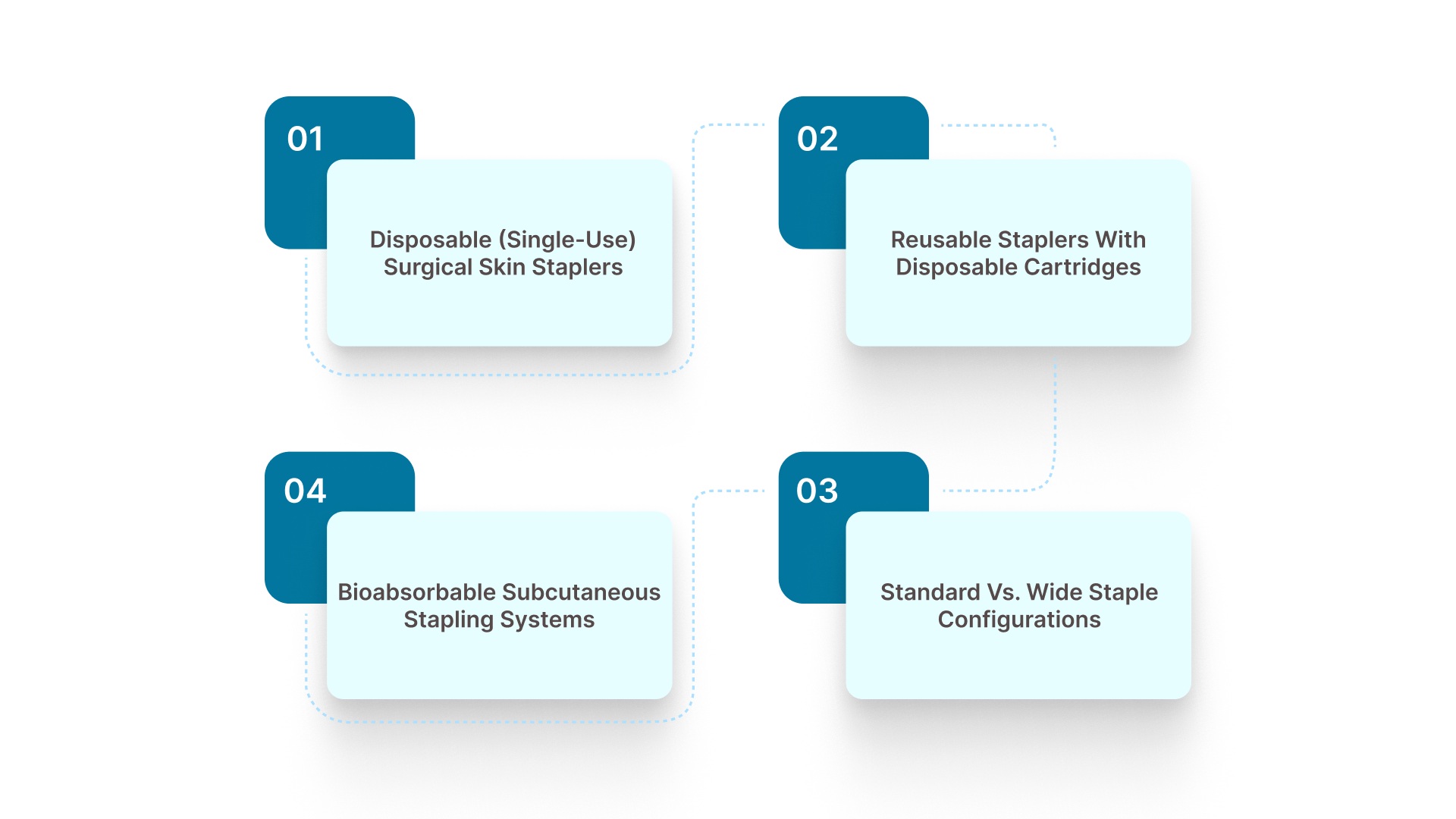
Selecting a surgical skin stapler depends on incision length, tissue thickness, and how closure fits into the overall OR (Operating Room) workflow. Each type supports a different balance between speed, control, and post-operative management.
1. Disposable (Single-Use) Surgical Skin Staplers
These are preloaded with metal staples and come sterile, ready for immediate use. They are the most commonly used option in the operating room due to their simplicity and consistent performance.
Key features:
- Preloaded and ready to use: Metal staples allow immediate deployment without setup and support faster closure at the end of procedures.
- No sterilization required: Eliminates reprocessing between cases, simplifying workflow and reducing turnover time.
- Consistent staple formation: Mechanical deployment produces uniform staple formation and spacing across the incision.
- Widely used across procedures: Common in general surgery, orthopedics, and OB-GYN for routine skin closure.
- Supports high-throughput settings: Designed for environments where closure time impacts overall OR efficiency.
2. Reusable Staplers with Disposable Cartridges
These systems use a reusable handle with replaceable staple cartridges. They are used in selected settings where cost control is a priority, but require additional sterilization and handling.
Key features:
- Reusable handle design: The main device is used across multiple procedures, reducing long-term equipment cost.
- Replaceable cartridges: Staple cartridges are changed per case based on procedural needs.
- Lower per-case cost over time: Can reduce device cost with repeated use, depending on volume and workflow.
- Requires sterilization: Adds reprocessing steps between cases, potentially affecting turnaround time.
- Operational coordination needed: Requires tracking of both reusable components and cartridges.
3. Standard vs. Wide Staple Configurations
Staple size selection is based on tissue thickness and wound tension. Choosing the correct configuration helps maintain stable wound approximation during healing.
Key features:
- Standard staples for routine use: Typically used for moderate tissue thickness and standard closures.
- Wide staples for high-tension areas: Used for thicker skin or areas such as the back, scalp, or joints where tension is higher.
- Supports tension distribution: Proper staple size helps maintain alignment without excessive pressure on tissue.
- Application varies by anatomical site: Selection depends on incision location and tissue characteristics.
- Impacts closure stability: Influences how securely the wound edges are held during early healing.
4. Bioabsorbable Subcutaneous Stapling Systems
These systems place fasteners beneath the skin rather than through it. Instead of external metal staples, they use bioabsorbable fasteners that hold the incision internally and are absorbed over time.
Key features:
- Subcutaneous placement: Fasteners are deployed under the skin, avoiding penetration of the outer skin surface.
- No removal required: Bioabsorbable material is naturally absorbed, eliminating the need for a removal procedure.
- No external metal staples visible: The absence of surface hardware changes post-operative management and patient experience.
- Reduces visible scarring: No external puncture marks associated with traditional metal staples.
- Simplifies post-operative workflow: Removes the need for a follow-up visit specifically for staple removal.
Examining the different types in use also highlights the limitations associated with traditional metal surgical skin staplers.
Key Limitations of Traditional Metal Surgical Skin Staplers
While a surgical skin stapler offers speed and consistency during closure, traditional metal staples come with trade-offs that directly impact post-operative workflow, patient experience, and overall case efficiency.
Below are the key limitations.

These limitations also highlight the process of staple removal and its impact on the overall surgical workflow.
What is Staple Removal and Its Impact on Surgical Workflow?
Staple removal is the postoperative step in which externally placed metal skin staples are removed once the incision has achieved sufficient initial healing.
This is typically performed around 7–14 days after surgery, depending on incision location, tension, and patient-specific healing factors.
The procedure involves using a sterile staple remover to deform each staple and lift it out of the skin without disrupting the incision. In some cases, alternate staples may be removed first to assess wound stability before complete removal.
Here’s what staple removal involves in practice:
- A scheduled follow-up visit: Removal must be performed within a defined timeframe based on the healing progress, and coordination is required afterward.
- A structured removal technique: Each staple is removed individually using a dedicated tool, with correct positioning to avoid tissue disruption.
- Wound assessment during removal: The incision is evaluated for healing, alignment, and stability before and during removal.
Here’s how the impact of staple removal on the surgical workflow:
- Extends closure beyond the operating room: Skin closure is not complete at the end of the procedure and requires a follow-up intervention to finalize.
- Adds coordination at scale: Managing removal visits increases outpatient workload, particularly in high-volume practices.
- Relies on timing and patient follow-through: Removal must occur within the appropriate window to support proper healing.
Once you understand how staple removal affects workflow, it also informs how practices evaluate and select the most suitable surgical skin stapler.
How to Choose the Right Surgical Skin Stapler for Your Practice?
Selecting a surgical skin stapler affects more than closure speed. It influences OR efficiency, postoperative workflow, and the consistency with which closure is performed across cases.
Here’s how you can choose the right surgical skin stapler for your practice:
- Match the device to incision type and length: Short incisions require precise placement in limited space, while longer incisions benefit from devices that support continuous, efficient closure.
- Consider tissue thickness and wound tension: Standard staples are suitable for routine closures, while wider staples are used in thicker skin or high-tension areas to maintain stable approximation.
- Balance closure speed with control: Staplers enable faster closure than sutures, while sutures may be preferred when fine adjustment of wound edges is required.
- Evaluate post-operative workflow impact: Metal staples require a follow-up removal step, which adds coordination and extends the closure process beyond the procedure.
- Account for procedure volume and OR efficiency: In higher-volume settings, faster closure can support more efficient case flow and reduce time spent on wound closure.
- Look beyond device cost to total impact: Device selection should consider staff time, follow-up visits, and coordination.
Final Thoughts
Closure decisions extend beyond the final step in the operating room, shaping overall case efficiency, post-operative coordination, and consistency across procedures. As surgical volume increases, the impact of closure time and follow-up requirements becomes more visible in day-to-day workflow.
In this context, SubQ It! SU-25 is designed for longer incisions, using 25 bioabsorbable fasteners to support closure of up to 25 cm while reducing closure time by approximately 25 minutes per case.
This is particularly relevant in high-volume or private practice settings where closure time directly influences throughput.
Contact us today to see how SubQ It! SU-25 can fit into your workflow and support more efficient, consistent closure.
FAQs
1. Can surgical skin staplers be used for all types of wounds?
No. They are typically used for clean, linear incisions where edges can be approximated easily. Irregular, contaminated, or high-precision wounds may require sutures or alternative closure methods.
2. Are surgical skin staplers used in pediatric procedures?
They can be used when appropriate, but the decision depends on incision type, skin thickness, and follow-up considerations. In some cases, alternative methods may be preferred to simplify postoperative management.
3. When can patients shower after surgical staples are placed?
Patients are usually advised to keep the incision dry for the first 24–48 hours. After that, showering may be allowed according to the procedure and the surgeon’s postoperative protocol.
4. Is anesthesia required for staple removal?
Staple removal is typically performed without anesthesia. Patients may experience brief discomfort or a pulling sensation at the incision site during the procedure.
5. Are dressings used after applying surgical skin staples?
Yes. Dressings are commonly applied after closure to protect the incision and support early healing. The type and duration depend on the procedure and wound characteristics.
6. What should be assessed before removing surgical staples?
The incision should be evaluated for adequate healing, proper edge approximation, and absence of infection or separation. In some cases, alternate staples may be removed first to confirm wound stability.


