
The surgical skin stapler price on your distributor's quote is easy to analyze. However, there are additional costs to consider, such as removal visits, additional OR minutes, and infection-related re-admissions.
A 2025 review of 18,268 vascular bypass procedures found that patients closed with staples developed surgical site infections at a higher rate than those closed with sutures. Downstream costs from that single complication can exceed the cost of the device itself.
This breakdown covers what staplers actually cost by type, what impacts the real price per case, and when bioabsorbable subcutaneous fit as a better option.
Key Takeaways
- Standard disposable metal skin staplers from major US distributors list between $10 and $15 per unit, while premium single-use models from specialty brands can exceed $40.
- Bioabsorbable subcutaneous skin staplers run $60 to $100 per device, with no removal visit required and no railroad-track scarring.
- The sticker price is the smallest component of total closure cost; infection, removal overhead, and OR time carry the real financial weight.
- One surgical site infection can add $30,689 to a single hospital admission, making every percentage point of infection risk a cost variable worth tracking.
- Surgeons who used absorbable dermal staples in breast reductions closed 21.8 minutes faster per case than those who hand-sewed, a gap that translates directly to OR cost savings.
- For private-practice surgeons who control purchasing, the total cost per case (device, OR time, removal, and complication risk) is a more accurate comparison metric than device cost alone.
What Is a Surgical Skin Stapler?
A surgical skin stapler is a handheld device that fires metal or polymer fasteners through or beneath the skin to close an incision. It replaces or supplements hand-sewn sutures during the final step of wound closure in the operating room.
Staplers fall into two broad categories. Traditional metal skin staplers place stainless steel staples through the epidermis. They're fast to fire, cost less per unit, and require a follow-up visit for removal in 7 to 10 days. Bioabsorbable subcutaneous (under the skin) staplers place polymer fasteners beneath the skin surface, where they dissolve over time with no removal needed.
The price spread between these categories is significant, and so are the downstream costs each one creates. Understanding both sides of that equation is what makes a surgical skin stapler price comparison useful rather than misleading.
What Does a Surgical Skin Stapler Cost in 2026?
Standard disposable metal skin staplers from major manufacturers list between $10 and $15 per unit through US medical supply distributors. Premium single-use models from specialty brands can run $40 or higher.
Here's how the main categories compare.
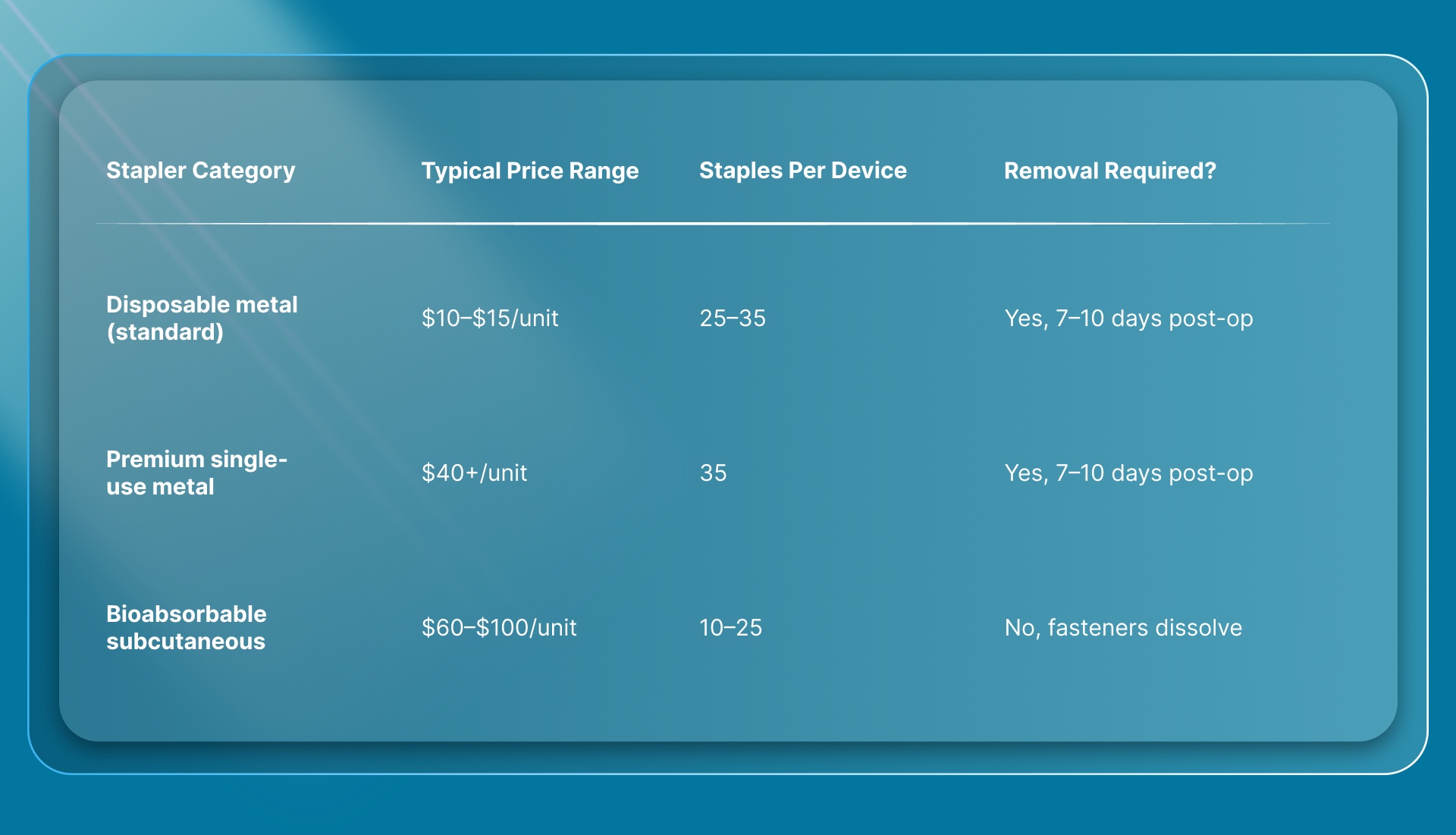
The wide range reflects differences in material, staple count, brand, and distribution channel. A $12 disposable and a $90 bioabsorbable device aren't competing on the same axis. One is priced to the device. The other is priced to the case.
The distinction matters more than the sticker number. A disposable skin stapler at $12 creates costs after the OR that a bioabsorbable option at $90 does not. The next question is what those costs actually look like.
What Factors Affect Surgical Skin Stapler Pricing?
Device price depends on a handful of procurement variables that every surgeon or purchasing manager already recognizes. None of these factors are complicated, but they stack.
Staple Count and Device Size
A 15-staple disposable costs less than a 35-staple version from the same manufacturer. Larger devices cover longer incisions in fewer passes, which justifies a higher price for the convenience. Matching staple count to incision length avoids overpaying for capacity you won't use.
Single-Use vs. Reusable Design
Single-use disposable staplers dominate the US market. Reusable skin staplers have a higher upfront cost but amortize across sterilization cycles. The trade-off is processing overhead (reprocessing, tracking, compliance) vs. per-unit savings.
Material and Mechanism
Metal stainless steel staplers are the lowest-cost option per unit. Bioabsorbable polymer devices cost more upfront because the fastener material is engineered to dissolve in tissue over weeks. The mechanisms also differ. Metal staplers fire through the skin surface, while subcutaneous devices place fasteners beneath it.
Purchasing Volume and GPO Contracts
Hospitals buying through group purchasing organizations typically get lower per-unit pricing than solo practices ordering from distributors. Bulk box pricing (12-packs, 24-packs) drops the per-unit cost by a few dollars compared to singles. A solo practice buying six units a month pays a different rate than a hospital system ordering hundreds.
Vendor and Distribution Channel
The same stapler model can list at noticeably different prices across distributors. Online medical supply retailers, manufacturer-direct sales, and GPO-contracted pricing all produce different numbers for the same SKU. Shopping the channel is standard practice, but comparing across channels without holding other variables constant creates misleading quotes.
Every one of these factors changes the line-item price. None of them changes the total cost per closure, which depends on what happens after the stapler fires.
How Does Device Price Compare to Total Closure Cost?
The device price is the easiest number on the invoice and the least useful one for evaluating real cost. Total closure cost includes the device, OR time consumed during closure, removal visit overhead (for metal staples), and complication-related expenses if the wound breaks down.
Removal visits add staff time, scheduling burden, supplies, and a patient slot that generates no procedural revenue. Every metal staple closure creates this downstream appointment. Bioabsorbable subcutaneous closure removes it from the workflow entirely.
OR time is one of the highest fixed costs in any surgical facility, and every additional minute of closure time adds to it. Published estimates for OR cost per minute vary widely depending on facility type, geography, and accounting method, with commonly cited averages landing in the $36 to $46 range. Even at the lower end of that spectrum, saving 15 to 20 minutes per closure creates a per-case reduction that adds up across a full surgical schedule.
The largest potential cost lives in complications. When a surgical site infection develops, the financial impact dwarfs any device price. A 2025 study across 38 US acute-care hospitals put the average per-admission penalty at $30,689 in added cost and 11.6 extra days in the hospital.
Looking beyond the initial admission, the picture gets worse. A claims-based analysis of commercial and Medicare Advantage patients found that total medical spending over 12 months ran $40,606 to $68,101 higher per SSI patient than per matched control, depending on the procedure.
A $12 metal stapler that contributes even a small incremental fraction of infection risk, removal cost, and added OR time can exceed the total case cost of a $60 to $100 absorbable wound closure device that requires no removal and closes faster. The denominator for a stapler price comparison should be the case, not the device.
How Do Metal Stapler Prices Compare to Bioabsorbable Options?
Metal skin staplers cost $10 to $15 per unit for standard disposables. Bioabsorbable subcutaneous fasteners run $60 to $100 per device. On sticker price alone, metal wins by a wide margin. In total case economics, though, the comparison changes.
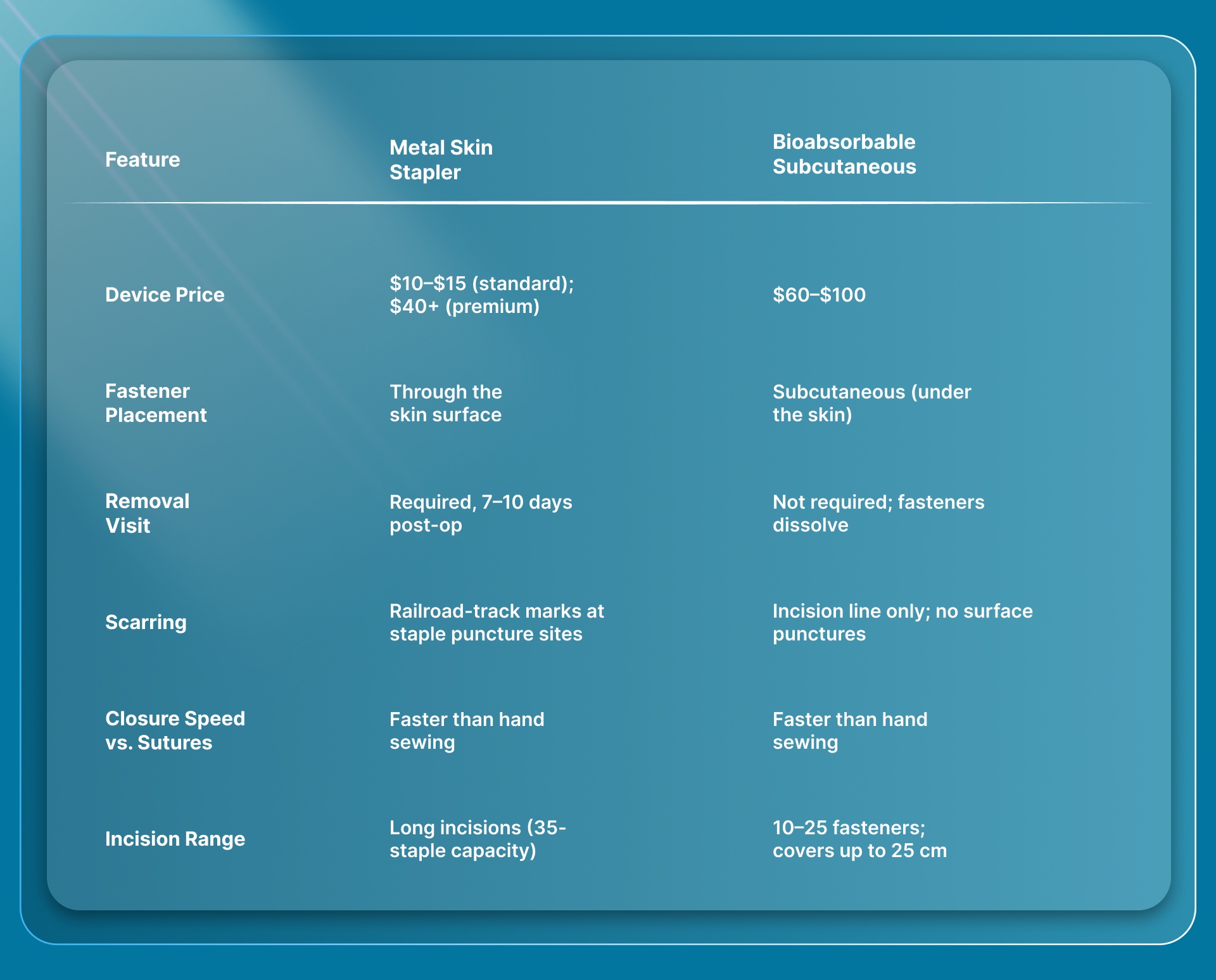
Metal staplers are faster than hand-sewn sutures, and that's a big advantage. Bioabsorbable subcutaneous staplers share that speed advantage while also removing the downstream cost of a removal appointment and the cosmetic liability of railroad-track scarring.
Columbia University researchers tracked 62 bilateral breast reductions and found that closure ran 21.8 minutes faster per case when surgeons used absorbable dermal staples instead of sutures. When the data were adjusted for tissue mass and pedicle type, the gap widened to 26.5 minutes, with no difference in complication rates between the two groups.
Bioabsorbable subcutaneous devices are available with 10 fasteners for incisions up to 10 cm, covering laparoscopic trocar ports and small incisions that surface-applied devices can't reach. Larger models carry 25 fasteners for incisions up to 25 cm. The category covers the same incision range that metal staplers serve, from 5 mm laparoscopic ports to open surgical closures.
The common objection to paying 5 to 8 times more per device is valid only if you're comparing device cost in isolation. Once you add the removal visit, OR time, scarring outcomes, and infection risk profile, the $60 to $100 bioabsorbable option frequently costs less per closure than the $12 metal alternative.
What Infection Risk Does Staple Closure Carry?
Metal staple closure has a documented association with higher surgical site infection rates in certain specialties. The clearest recent data comes from vascular surgery.
Researchers reviewed 18,268 lower extremity bypass cases logged in the Vascular Quality Initiative between 2014 and 2024. Stapled incisions developed infections at a rate of 3.46% vs. 2.06% for sutured incisions, translating to 57% higher adjusted odds of SSI in the staple group. Staple closure was also tied to 30% higher odds of returning to the OR.
These figures come from vascular surgery, and infection risk varies by procedure type, patient population, and wound classification. The finding doesn't mean every metal staple closure will produce an SSI. It does mean that the closure method contributes to infection probability, and that probability has a financial cost attached to it.
The SSI risk profile belongs in the price comparison alongside device cost and removal overhead. A bioabsorbable subcutaneous approach avoids surface punctures entirely, which removes one of the mechanical pathways through which skin-surface staplers may introduce contamination.
Infection-related costs aren't hypothetical budget items. They're documented per-admission charges that can reach five figures on a single case. Factoring them into a stapler price evaluation doesn't inflate the comparison. It completes it.
How Should Surgeons Evaluate Stapler Cost for Private Practice?
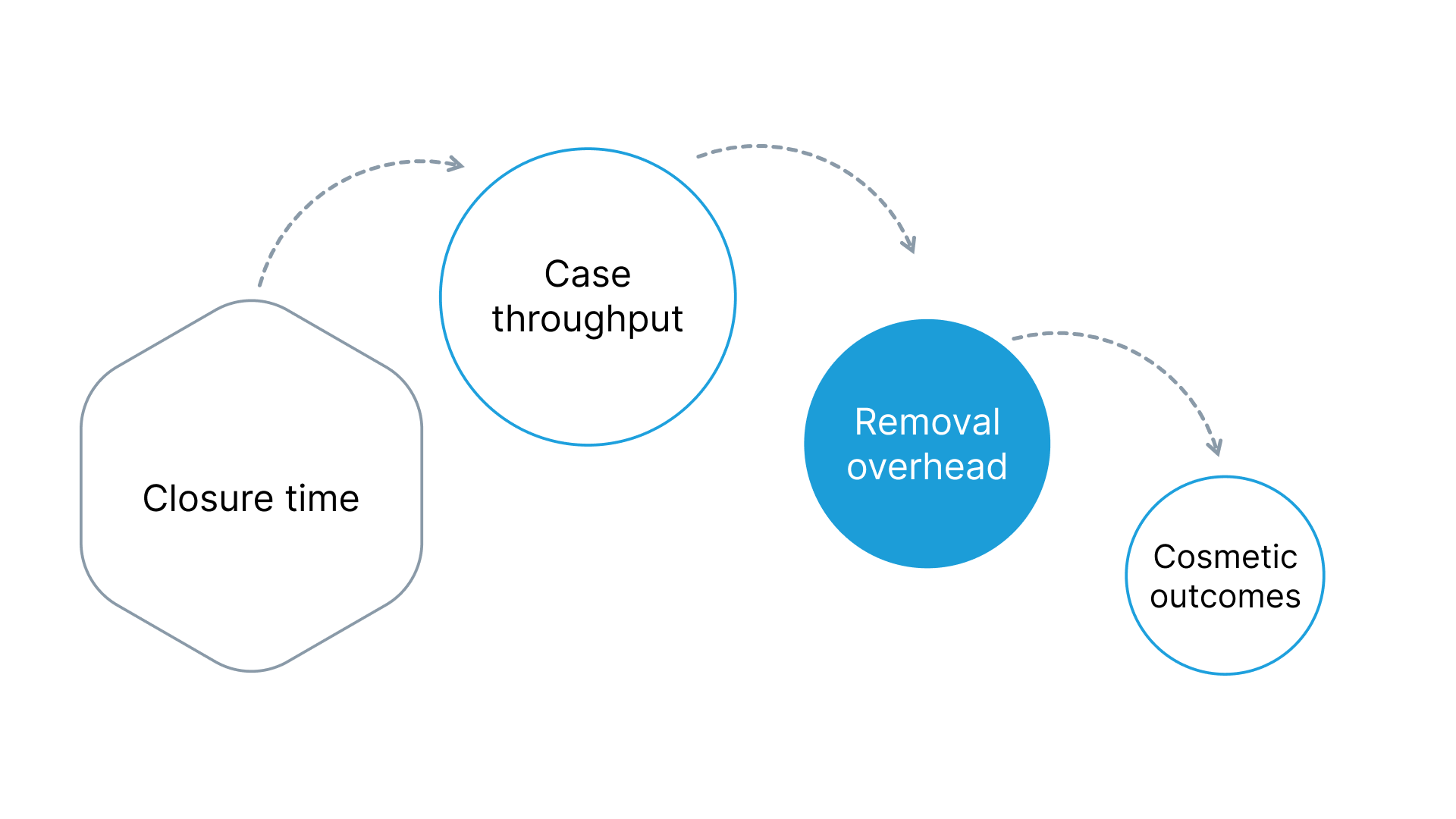
For a surgeon who owns the practice, the useful comparison metric isn't device cost per unit. It's the total closure cost per case, measured against the revenue that freed-up OR time can generate.
Four cost components deserve attention when comparing staplers for a private surgical practice.
- Closure time: Absorbable dermal staples have been shown to cut closure time by roughly a third compared to hand-sewn sutures in plastic surgery cases. At commonly cited OR cost estimates ($36 to $46 per minute), even 15 to 20 minutes saved per case adds up across a full week of procedures.
- Case throughput: Twenty-plus minutes recovered per closure creates room in the schedule. In a private cosmetic or plastic surgery practice running four to six procedures a day, that recovered time can open capacity for additional cases without extending the day. Each additional case generates revenue that far exceeds a $60 to $100 device.
- Removal overhead: Every metal staple closure generates a follow-up appointment that occupies a staff member, consumes a patient slot, and creates liability if the patient delays or misses the visit. Bioabsorbable subcutaneous fasteners dissolve on their own. The follow-up disappears from the schedule permanently.
- Cosmetic outcomes: For plastic and cosmetic surgeons, scarring from staples used for closure is a liability conversation with every patient. Railroad-track marks from metal staples are visible and permanent. Subcutaneous placement leaves only the original incision line.
Private-practice surgeons who evaluate staplers on device cost alone will consistently choose the $12 option. Surgeons who evaluate on total cost per case, including OR time, removal overhead, complication risk, and cosmetic outcomes, consistently land on a different answer.
Where Can Surgeons Buy Surgical Skin Staplers?
Purchasing channels for surgical skin staplers in the US fall into four categories.
- Manufacturer direct: Some companies sell directly to practices and hospitals. Bioabsorbable subcutaneous staplers, for example, are often available direct from the manufacturer, which simplifies quoting and removes distributor markup.
- Medical supply distributors: Companies like McKesson, Medline, and Cardinal Health carry disposable metal staplers through their standard catalogs. Pricing varies by account volume.
- Group purchasing organizations (GPOs): Hospitals and health systems with GPO contracts get negotiated pricing on commodity devices, including metal skin staplers. Solo practices and small ASCs typically don't have GPO access for every product category.
- Online medical retailers: Distributors like Vitality Medical, Tiger Medical, and WestCMR list staplers online with visible per-unit pricing. These are useful for price benchmarking, though not every listed SKU is available at the quoted price in all regions.
For standard disposable metal staplers, the supply chain is mature and commoditized. Pricing differences across channels are usually a few dollars per unit. For bioabsorbable subcutaneous staplers, going direct to the manufacturer is the most common path and the one that gives you the clearest pricing and product support.
If you're evaluating wound closure device companies for a new product category, request quotes that include per-unit device cost and ask about volume-based pricing tiers. Compare those quotes against the total cost per case, not against the per-unit cost of a metal stapler in a different category.
Final Thoughts
The surgical skin stapler price that shows up on an invoice is the beginning of the cost conversation, not the end of it. Removal visits, OR time, complication risk, and scarring outcomes all contribute to total closure cost per case. When those factors enter the comparison, the cheapest device per unit is often the most expensive option per procedure.
For private-practice surgeons comparing closure methods, the question worth asking is what each case costs from incision close through final follow-up. Run the math on your own case mix, OR cost structure, and complication rates. The answer will be specific to your practice, and it will almost certainly be different from the device price alone.
SubQ It! bioabsorbable subcutaneous fasteners dissolve on their own, require no removal visit, and leave no railroad-track scarring. Every week a practice continues using metal staples for closures that a subcutaneous approach handles better is a week of added removal appointments, avoidable OR minutes, and cosmetic results that fall short of what's possible. Contact us today to request a custom quote.
FAQs
1. How Many Staples Come in a Standard Surgical Skin Stapler?
Standard disposable metal skin staplers typically carry 35 staples per device. The exact count depends on the manufacturer and model, so check the product specifications before ordering to match the staple count to your expected incision length.
2. Do Bioabsorbable Staples Cost More Than Metal Staples Per Unit?
Yes, bioabsorbable subcutaneous skin staplers cost more per device than standard disposable metal models. The higher unit price reflects the polymer material, subcutaneous placement mechanism, and the fact that no removal appointment or remover tool is needed afterward.
3. Are Surgical Skin Stapler Prices Covered by Insurance Reimbursement?
Yes, surgical skin staplers are generally reimbursable as part of the overall procedure cost. The device expense is typically bundled into the facility fee rather than billed as a separate line item, so the reimbursement structure depends on the procedure code and payer contract.
4. Can One Surgical Skin Stapler Be Used on More Than One Patient?
No, single-use disposable surgical skin staplers are designed for one patient only and must be discarded after the procedure. Reusable models exist and can be sterilized between patients, though they require tracking and reprocessing infrastructure.
5. Are Reusable Surgical Skin Staplers More Cost-Effective Than Disposable?
Reusable staplers can lower per-unit cost over time if case volume is high enough to justify the upfront investment and reprocessing workflow. For lower-volume practices, the savings often don't offset the sterilization, tracking, and compliance overhead.
6. Does Staple Width or Gauge Affect Surgical Skin Stapler Price?
Yes, wider-gauge staples designed for thicker tissue or high-tension closures generally cost more per device than standard-width models. The premium reflects the heavier-gauge wire or polymer and the modified firing mechanism required for deeper placement.
About SubQ It!
SubQ It! manufactures bioabsorbable subcuticular closure systems that combine the speed of staplers (7 seconds per fastener) with subcuticular placement. With FDA clearance (K131563) and ISO 13485 certification, the system eliminates staple removal and delivers closure speeds 7X faster than manual sutures.
For more information, contact the team at sales@subq-it.com or call +1 339.933.8811.


