Disposable skin staplers have looked roughly the same for years. A pre-loaded metal device, a squeeze handle, and a row of stainless-steel staples pressed through the skin surface.
What has changed, though, is the arrival of bioabsorbable subcutaneous staplers that place fasteners under the skin instead of through it. Skin closure dropped to nearly 16.2 minutes with an absorbable stapler in a 4,311-patient mastectomy series, compared to almost 36.5 minutes by hand, and infection rates were identical between groups.
This post covers both sides of the disposable skin stapler category so you can evaluate which type matches your clinical priorities and case volume.
Key Takeaways
- A disposable skin stapler is a sterile, pre-loaded, single-use device designed for wound closure, and the category now includes both metal surface staplers and bioabsorbable subcutaneous options.
- Metal surface staplers are fast (saving 0.30 to 5.50 minutes over sutures) but require removal in 7 to 10 days and leave railroad-track scars at each puncture site.
- Bioabsorbable subcutaneous staplers place fasteners under the skin that dissolve naturally, with no removal visit and no surface puncture marks.
- Infection rates between metal staples and sutures show no significant difference in the largest published meta-analysis (42 RCTs, 11,067 patients), meaning the closure choice comes down to scarring, time, and follow-up logistics.
- Surface staplers can't effectively close very small laparoscopic trocar-site incisions (a few millimeters), while bioabsorbable subcutaneous devices cover incisions from a few mm up to 25 cm.
What Is a Disposable Skin Stapler?
A disposable skin stapler is a sterile, pre-loaded, single-use wound closure device designed to close surgical incisions quickly during or after a procedure. Unlike reusable skin staplers that require sterilization between uses, disposable models come ready to fire and get discarded after a single case. The single-use design reduces cross-contamination risk and removes the need for reprocessing between patients.
The category has traditionally meant one thing. A handheld device loaded with stainless-steel staples that pierce the skin surface and bend closed underneath. These metal surface staplers remain the most widely stocked option in ORs across the United States. They're reliable, they're fast, and surgeons have used them for decades.
What's newer in the category is the bioabsorbable subcutaneous stapler, a wound closure system that places polymer-based fasteners under the skin rather than through it. These fasteners dissolve over time, so there's no removal step and no surface entry marks.
How Do Metal Surface Staplers Compare to Bioabsorbable Subcutaneous Staplers?
The two types differ on placement, material, removal requirements, cosmetic outcomes, and the incision sizes they can serve. Here's how they line up.
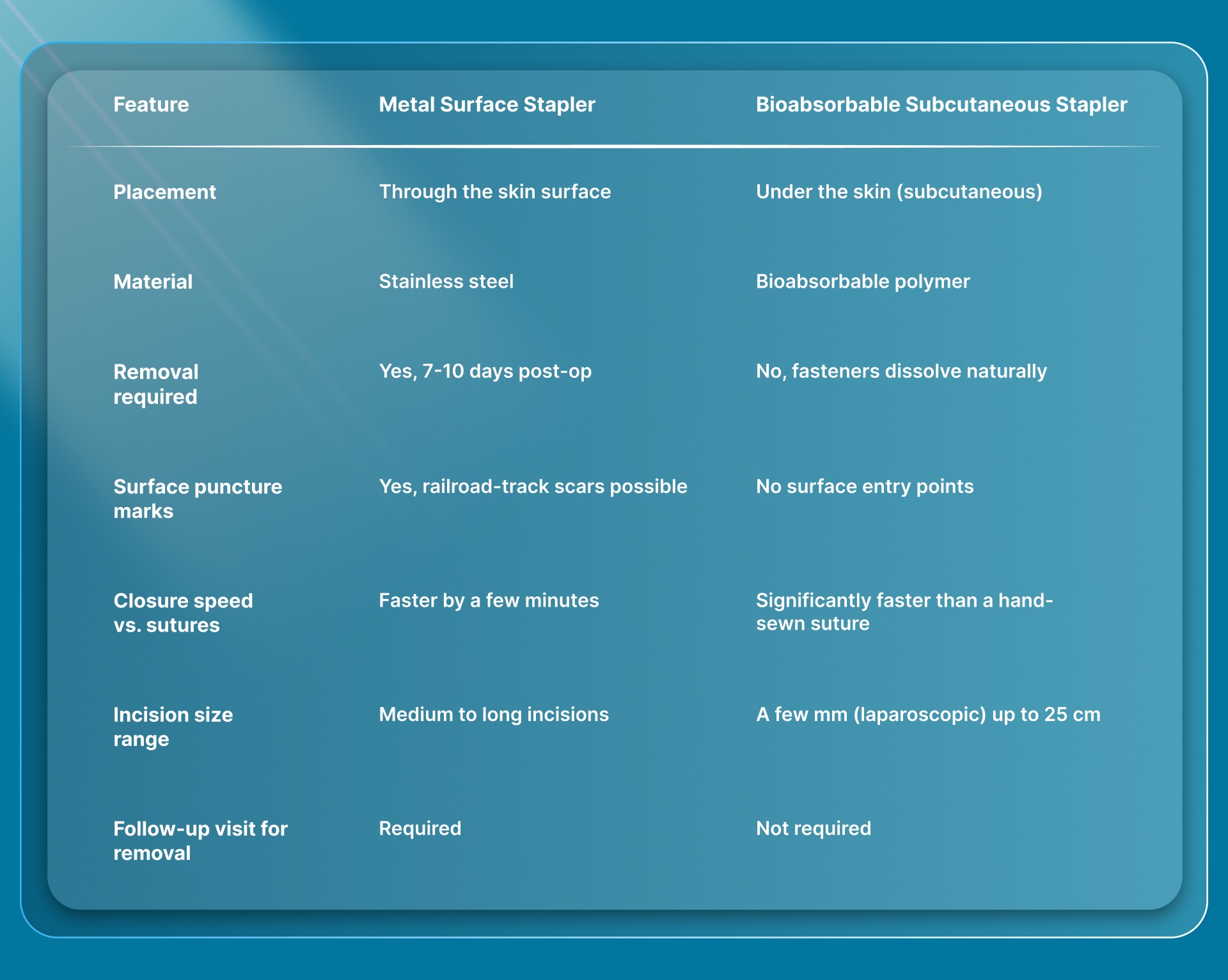
Each stapler type has a clear clinical profile. Metal surface staplers close fast and cost less per unit, making them a familiar choice for straightforward incisions where cosmetic outcome is a lower priority. Bioabsorbable subcutaneous staplers add clinical value where scarring matters, where follow-up visits create logistical burden, or where the incision is too small for a surface device to reach.
Scarring Outcomes
Metal staples leave paired puncture marks on either side of the incision line. These railroad-track scars become more visible the longer staples stay in place. In orthopedic wound data, scars at staple insertion sites were measurably wider than suture-closed scars, and the marks grew more pronounced when removal was pushed past 10 days. Across 3,705 abdominal surgery patients, patients consistently rated their cosmetic results and overall satisfaction higher when sutures were used instead of staples.
Bioabsorbable subcutaneous staplers avoid this issue entirely. The fasteners go under the skin, so there are no surface puncture sites, no insertion marks, and no external hardware to create scarring pressure points. The only visible mark is the original incision line itself.
Infection Rates
Infection data doesn't clearly favor one surface closure method over another. Across 42 RCTs and 11,067 patients, suture-closed wounds developed infections at a rate of 4.90%, while staple-closed wounds came in at 6.75%. The difference between those numbers wasn't statistically significant. Mayo Clinic's review of 18,842 posterior spine cases reinforced that finding, with SSI rates holding at 0.8% regardless of whether the surgeon used staples or sutures.
For bioabsorbable subcutaneous staplers, infection rates have tracked similarly. The Lee et al. mastectomy series showed 0.38% infection with an absorbable stapler and 0.36% with hand-sewn suture. A cesarean delivery cohort of 31,419 patients confirmed the same pattern, with wound infection rates holding steady whether surgeons used absorbable subcutaneous staples, metal staples, or sutures.
Removal Requirements
Metal surface staplers require a scheduled removal visit, typically 7 to 10 days after surgery. This consumes a clinic appointment, involves a dedicated stapler remover tool, and can be uncomfortable for the patient. For busy practices, each removal visit occupies time that could serve a new consultation or follow-up for a different case.
Bioabsorbable subcutaneous staplers skip this step entirely. The fasteners dissolve in place, so there's no return visit, no removal instrument, and no scheduling coordination with the patient. This is a direct efficiency gain for both the practice and the patient.
What Incision Sizes Can Each Stapler Type Handle?
Metal surface staplers perform well on medium to long incisions where the wound edges are accessible, and the skin surface provides enough real estate for staple placement. For shorter or smaller incisions, particularly the trocar-port sites common in laparoscopic procedures, surface staplers aren't practical. The wound aperture is simply too small.
This is a recognized challenge in laparoscopic surgery. A 2024 review in Cureus noted that port-site incision closures are technically difficult because of the small wound aperture, and incomplete fascial closure at these sites is common. European and American Hernia Society guidelines recommend fascial closure for trocar sites 10 mm and larger, but effective skin closure on these tiny incisions remains a device-level limitation for surface staplers.
SubQ It! addresses this range limitation with two variants. The SubQ It! SU-10 carries 10 bioabsorbable fasteners and closes incisions up to 10 cm, including very small laparoscopic stapler trocar-port sites where surface devices can't operate. The SubQ It! SU-25 carries 25 fasteners and handles incisions up to 25 cm, covering longer closures in open surgery, plastic surgery, and other specialties. Between the two, the system works across the full spectrum of incision sizes surgeons encounter.
What Are the Risks and Limitations of Disposable Skin Staplers?
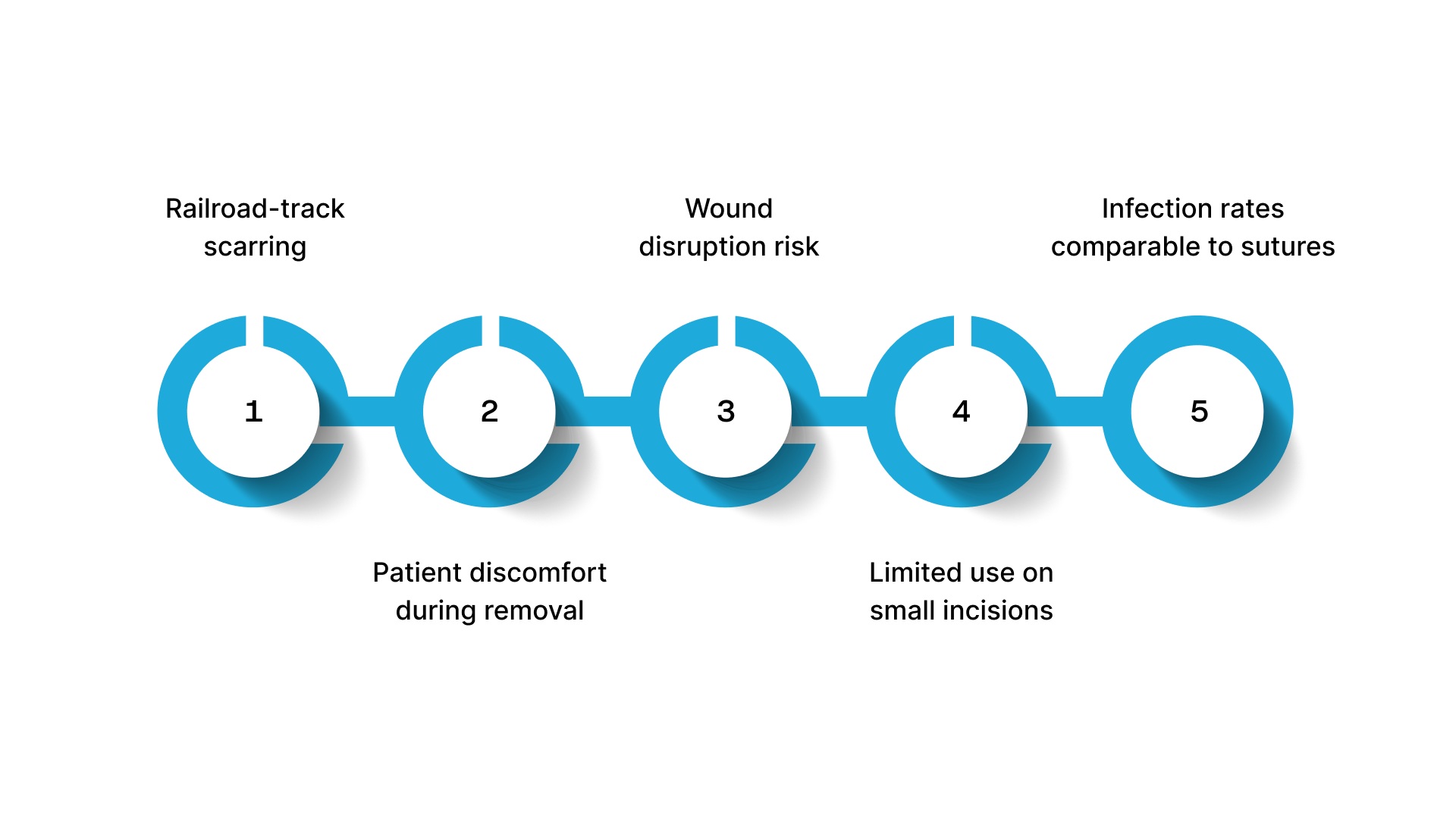
No closure device is without tradeoffs. Disposable metal surface staplers carry specific limitations worth weighing against your case mix and patient expectations.
- Railroad-track scarring: Surface staples leave paired puncture marks that are cosmetically distinct from the incision line. Delayed removal makes these marks more prominent.
- Patient discomfort during removal: Staple removal is a procedural step that patients frequently describe as painful, and it requires a staple pin remover instrument and a dedicated clinic visit.
- Wound disruption risk: When 300 cesarean patients were randomized across closure methods, wounds closed with metal staples broke down more often than those closed with sutures. By 4 to 6 weeks, absorbable subcuticular sutures produced the best cosmetic results of all three groups.
- Limited use on small incisions: Surface staplers require enough wound surface area to accommodate staple placement. Very small trocar-port incisions aren't accessible for these devices.
- Infection rates comparable to sutures: Large datasets consistently show no meaningful difference in surgical site infection rates between staples and sutures, so infection risk alone doesn't favor or penalize metal staplers as a closure choice.
How Does Stapler Choice Affect OR Time?
Closure time varies significantly depending on the device and the comparison method. Metal surface staplers close faster than sutures, and bioabsorbable subcutaneous staplers close faster than hand-sewn subcuticular closure by an even wider margin.
How much faster? According to a study of non-obstetric surgery, the speed advantage ranged from 0.30 to 5.50 minutes per case, depending on the procedure. That's a modest gain, and it shrinks further on shorter incisions.
The more striking time difference appears when bioabsorbable subcutaneous staplers replace hand-sewn suture. Skin closure time in mastectomy patients dropped to 16.2 minutes with an absorbable stapler, compared with 36.5 minutes by hand in a 4,311-patient series. That's 20 minutes back per case. Bilateral breast reductions followed a similar pattern, with total procedure time falling from 170 minutes to 154 minutes after switching from suture-only closure to an absorbable dermal stapler, and complication rates stayed the same.
For a private practice surgical skin stapler workflow, those minutes add up. Each minute of OR time carries a published benchmark cost of $37.45 in the inpatient setting (FY2014 data from 302 California hospitals). A 20-minute reduction in closure time directly increases OR availability for additional cases, particularly in high-volume settings where throughput determines revenue.
What Should Surgeons Evaluate Before Choosing a Disposable Skin Stapler?
The right stapler depends on your case mix, your patient population, and how your practice handles follow-up. There's no universal answer, but there are specific criteria that separate a good fit from a poor one.
Case Mix and Incision Size Range
If your cases span from laparoscopic ports to open incisions, you need a device that works across that range. Metal surface staplers handle medium and long incisions well, but can't close very small trocar sites. A bioabsorbable subcutaneous system like SubQ It! covers incisions from a few millimeters (SU-10) up to 25 cm (SU-25), reducing the need to switch between closure methods mid-case or across cases.
Cosmetic Outcome Priority
For patients where scarring is a primary concern (plastic surgery, cosmetic procedures, visible incision sites), the closure device directly affects the result. Metal staples leave surface puncture marks. Subcutaneous fasteners don't. If your practice markets on skin closure outcomes and patient satisfaction, that difference carries weight.
Follow-Up Workflow
Every metal staple case generates a downstream removal appointment. For high-volume practices, those removal slots accumulate. If your schedule is already tight, a closure method that skips the removal visit frees clinic capacity without reducing care quality.
Cost Per Case vs. Total Practice Cost
Metal disposable staplers have a lower unit price, but the total cost includes the removal visit, the remover instrument, and the clinic time consumed. Bioabsorbable subcutaneous staplers have a higher upfront cost per device, but they eliminate follow-up costs entirely. The net calculation depends on how you value clinic time and how many staple cases your practice runs per week.
Are Bioabsorbable Subcutaneous Staplers a Better Option Than Metal Surface Staplers?
For surgeons who prioritize cosmetic results, fewer follow-up visits, and OR efficiency, bioabsorbable subcutaneous staplers address the three persistent drawbacks of metal surface devices. There's no removal visit, no railroad-track scarring, and the time savings over hand-sewn suture are substantial.
The evidence supports this across large datasets. In the cesarean cohort of 31,419 patients, absorbable subcutaneous staples matched or outperformed metal staples and sutures on surgery time, hospitalization length, and infection rates. In the mastectomy series, the absorbable stapler cut closure time by more than half compared to hand-sewn suture. In breast reduction cases, procedure time dropped with no increase in complications. These aren't niche findings from small pilot studies. They're large-cohort, real-world results.
The tradeoff is a higher per-unit device cost. In metal-stapler cases where cosmetic outcome is a lower priority, and the removal visit isn't logistically burdensome, a traditional surface stapler may still make sense. Where scarring matters, where follow-up creates scheduling friction, or where the case mix includes very small sutureless skin closure incisions that surface devices can't reach, bioabsorbable subcutaneous staplers are the stronger clinical and economic choice.
Final Thoughts
The disposable skin stapler category is broader than the product listings on page one of Google suggest. Metal surface staplers close quickly and cost less per unit, but they create a downstream burden in removal visits and cosmetic liability in railroad-track scars. Bioabsorbable subcutaneous staplers solve both of those problems while maintaining comparable infection profiles and delivering meaningful OR time savings over hand-sewn closure.
For surgeons evaluating which stapler fits their practice, the decision comes down to what you're closing, who you're closing for, and whether your current workflow accounts for the full cost of follow-up. The data across mastectomy, cesarean, and breast reduction cohorts point in the same direction. Subcutaneous bioabsorbable closure is safe, efficient, and cosmetically superior to metal surface staples.
SubQ It! is a bioabsorbable wound closure system that places subcutaneous (under the skin) fasteners across incisions from a few millimeters to 25 cm. The fasteners dissolve naturally, leaving no railroad-track marks and requiring no removal visit. Surgeons still using metal surface staplers across their full case mix are scheduling removal appointments and accepting cosmetic tradeoffs that a subcutaneous option can avoid. Contact us today to request a custom quote.
FAQs
1. Can Disposable Skin Staplers Be Used on Pediatric Patients?
Yes, disposable skin staplers can be used on pediatric patients when the incision site and tissue thickness are appropriate for staple placement. The surgeon's clinical judgment on wound tension and skin quality determines whether staples or an alternative closure method is the better choice for each pediatric case.
2. Do Disposable Skin Staplers Require Special Storage Conditions?
No, most disposable skin staplers are stored at room temperature in their sterile packaging until use. Manufacturers specify storage guidelines on the packaging, and as long as the sterile barrier remains intact, the device stays ready for use without refrigeration or controlled environments.
3. How Many Staples Does a Typical Disposable Skin Stapler Hold?
Pre-loaded staple counts vary by manufacturer and model, with common configurations ranging from 15 to 35 staples per device. The number you need depends on incision length, staple spacing, and whether you're closing one incision or several port sites in the same case.
4. Can a Disposable Skin Stapler Be Used on the Face?
No, surface metal skin staplers are generally avoided on the face because of the high cosmetic sensitivity of facial skin. For facial incision closure, surgeons typically choose fine sutures or tissue adhesives that offer better control over scar appearance in visible areas.
5. What Happens if a Metal Skin Staple Is Left in Too Long?
Delayed removal increases the visibility of staple insertion marks and raises the risk of tissue overgrowth around the staple legs. Leaving staples in beyond the recommended 7 to 10 days can make removal more difficult and worsen the cosmetic outcome at the staple entry sites.
6. Do Bioabsorbable Staplers Work Through Thick Skin?
Yes, bioabsorbable subcutaneous staplers are designed to work in subcutaneous tissue across a range of skin thicknesses. Proper tissue eversion and wound edge alignment are critical for secure fastener placement, regardless of the patient's skin thickness or body habitus.