
A 2024 observational study watched 147 cases in seven pediatric ORs and found instrument problems in 34.37% of them. Every affected case lost an average of nearly 10.16 minutes to missing, broken, or malfunctioning tools. For a high-volume practice, those minutes add up fast.
Disposable scalpel blades bypass the reprocessing chain entirely. There's no sterilization cycle, no tray assembly error, and no chance of grabbing a blade that's lost its edge from repeated autoclaving. The trade-off is a per-unit cost, but in many surgical settings, that cost runs lower than the reprocessing burden it replaces.
This post covers the types, sizes, safety features, and practical selection criteria for disposable scalpel blades, for surgeons evaluating single-use options in 2026.
Key Takeaways
- Disposable scalpel blades are single-use surgical cutting instruments that arrive factory-sterile and pre-sharpened, removing reprocessing from the workflow entirely.
- Blade sizes follow a standardized numbering system (#10, #11, #15, #20, and others) where each number corresponds to a specific shape and surgical application.
- Sharps injuries among OR staff have a pooled self-reported prevalence of 41.5%, and safety-engineered disposable scalpels with retractable or shielded blades are designed to reduce exposure to them.
- Instrument reprocessing errors affected 34.37% of surgical cases in a 2024 study, with delays averaging 10.16 minutes per affected case.
- Disposable blades offer a consistent factory-sharp edge that doesn't degrade with repeated sterilization, which matters for incision quality and downstream wound healing.
What Is a Disposable Scalpel Blade?
A disposable scalpel blade is a single-use surgical cutting instrument intended for one use and then discarded. Unlike traditional setups where a reusable handle (#3 or #4) accepts a detachable blade that's swapped between cases, fully disposable scalpels arrive factory-sterile with the blade already integrated into a lightweight handle.
Some disposable scalpels use a fixed-blade design, while others feature a retractable or shielded blade to prevent sharp injuries. Both types skip the sterilization and tray assembly steps required for reusable handles. The blade arrives sharp, stays sharp throughout one procedure, and goes straight into a sharps container after use.
The shift toward single-use surgical instruments has accelerated in recent years, particularly in ambulatory surgery centers and private practices where reprocessing infrastructure is limited. In fact, in a 2025 study, researchers used borescopes to peer inside the channels of reusable surgical instruments and found leftover debris inside every single one inspected, with contamination persisting even after instruments went through multiple rounds of cleaning.
What Are the Standard Disposable Scalpel Blade Sizes and Numbers?
Disposable scalpel blades follow the same standardized numbering system used across all surgical scalpels. Each number identifies a specific blade shape, curvature, and cutting profile matched to particular tissue types and procedures.
Here's how the most common blade numbers break down:
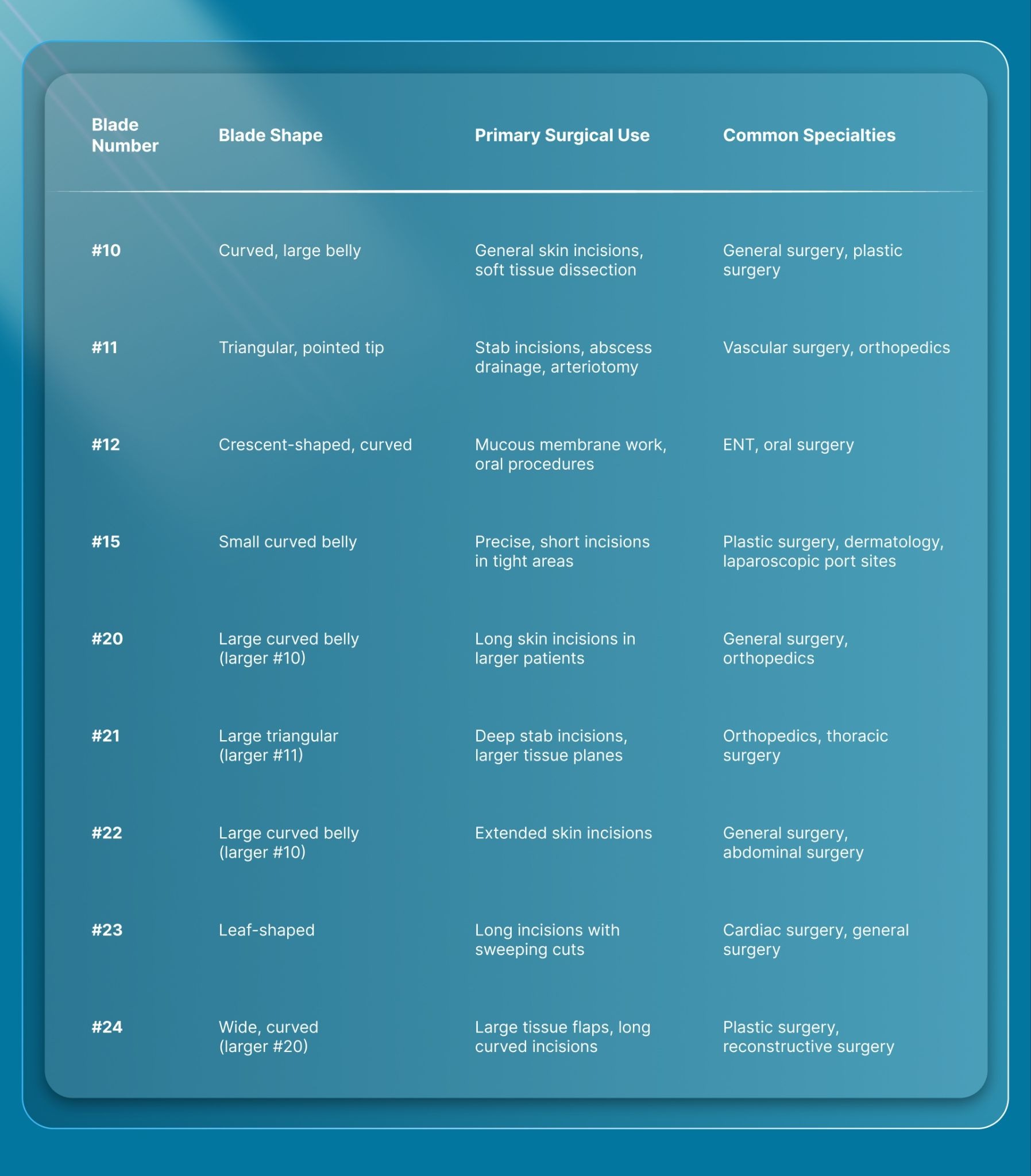
Smaller blades (#10, #11, #12, #15) pair with the #3 handle, while larger blades (#20, #21, #22, #23, #24) pair with the #4 handle. In a fully disposable scalpel, the handle comes pre-attached, so the pairing is already set by the manufacturer.
The #10 and #15 are the two most widely used blades in surgical practice. The #10 handles most general incisions, while the #15 is the go-to for fine, controlled cuts where precision matters more than reach. For sterile scalpel work in laparoscopic port site creation, the #15 or #11 is a common choice.
How Do Disposable Scalpel Blades Compare to Reusable Handle Systems?
Disposable scalpel blades and reusable handle-plus-blade systems achieve the same clinical task, but they differ in how they get there. The practical differences show up in sterility assurance, edge consistency, safety profile, and workflow burden.
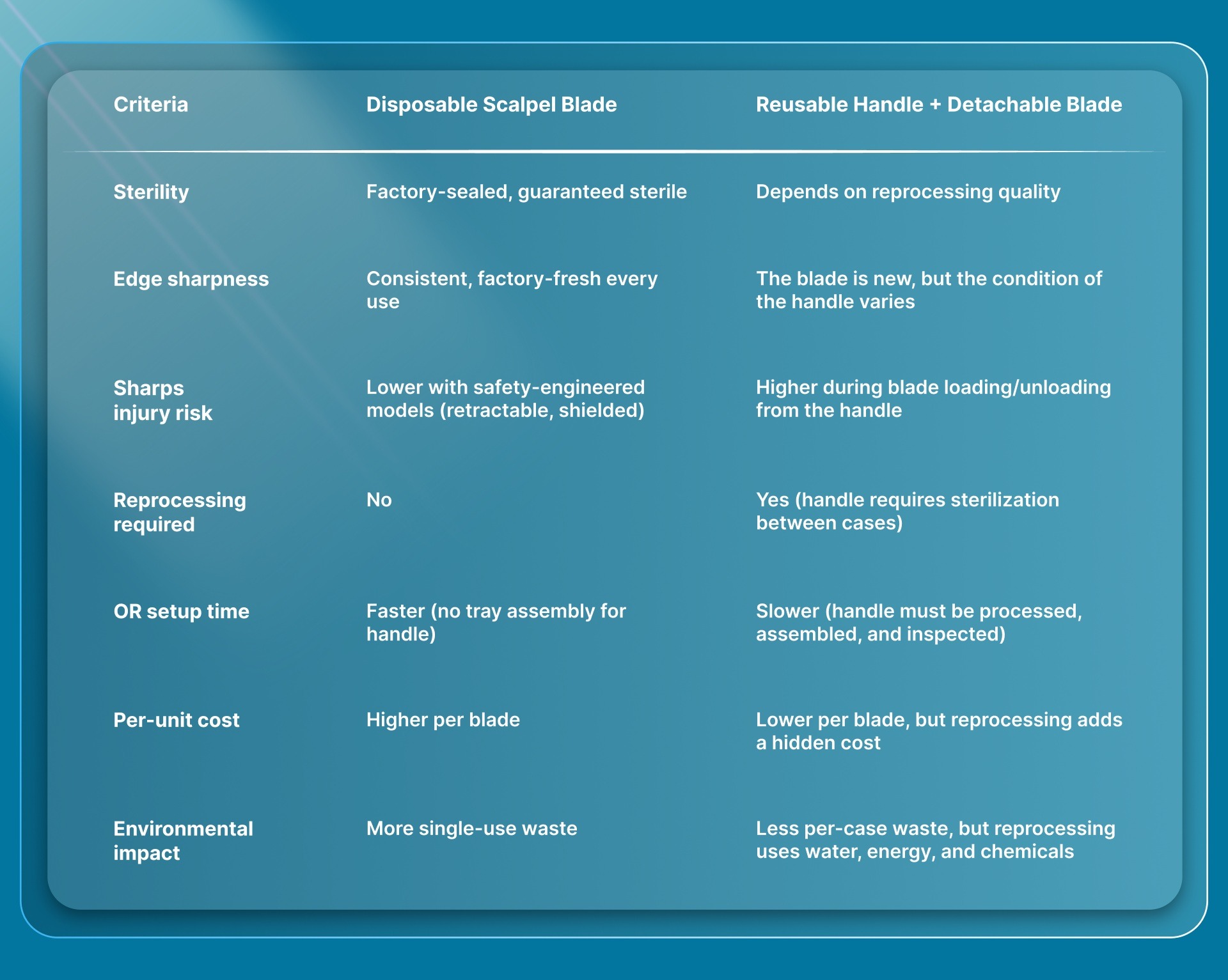
A 2025 review on single-use surgical equipment flagged a troubling pattern: infectious outbreaks have been linked to reprocessed devices even at hospitals that followed every recommended sterilization step. Cutting down the number of reusable instruments on each tray can meaningfully reduce sterilization workload and cost.
For high-volume practices and ambulatory surgery centers, the reprocessing burden often tips the equation toward disposable options. A 2024 observational study watched 147 cases unfold across seven ORs and found that something went wrong with the instruments in 34.37% of them. The most common cause was a breakdown in visual inspection, which drove 88.6% of all errors. Disposable blades remove the inspection, sterilization, and tray assembly steps from the workflow entirely.
The trade-off is environmental. Single-use instruments generate more waste per case. Practices weighing this decision should consider whether the sterility assurance, time savings, and safety profile of disposable blades justify the environmental cost for their surgical volume and case mix.
Why Are Sharps Injuries Still a Problem in the Operating Room?
Sharps and needlestick injuries are common in the OR. A 2023 meta-analysis that combined data from 16 studies put the self-reported injury rate among surgical staff at 41.5%. About 22% of those injuries happened during instrument hand-offs between team members.
The trend hasn't improved over time. NIOSH tracked sharps injury data across 89 hospitals in Massachusetts from 2002 through 2020. ORs and procedure rooms led every year. Other clinical areas saw their numbers fall, but the surgical setting stayed flat.
Across all US hospitals, sharps injuries add up to roughly 385,000 cases every year. The operating room drives a disproportionate share of that number. One 2023 occupational analysis found that working in a surgical setting multiplies the sharps injury risk by 16 times compared to the general workforce, and that younger clinicians between 25 and 34 are injured most often.
The numbers are still climbing. EPINet logged 1,945 sharps injury reports in 2024, a 10% increase over the 1,687 reported the year before. Splash and splatter exposures rose 15% in the same window.
Scalpel blades are among the most common sharps involved in OR injuries. Safety-engineered disposable scalpels with retractable or shielded blades are designed to reduce this exposure by enclosing the blade after use. OSHA's Bloodborne Pathogens Standard requires employers to evaluate and adopt safer sharps devices where feasible, and disposable scalpels with built-in safety features directly address that requirement.
For practices tracking sharps injury data or facing OSHA compliance reviews, switching to safety-engineered disposable blades is one of the most straightforward interventions available. The blade is used, retracted, or shielded, and discarded without ever being passed exposed across the field. When choosing surgical scissors and other sharps for the OR tray, the same safety-first logic applies.
What Should Surgeons Consider When Choosing a Disposable Scalpel Blade?
Selecting the right disposable scalpel blade depends on the procedure, the tissue type, the safety profile you need, and how the incision fits into your overall closure plan. Here are the factors that matter most.
Blade Geometry and Procedure Match
The blade number determines the shape, and the shape determines what the blade does well. Matching the blade to the procedure reduces tissue trauma and creates a cleaner wound edge for closure.
- #15 for precision work: Fine control for small incisions in plastic surgery, dermatology, or laparoscopic port creation.
- #10 for general incisions: The most common choice for standard skin incisions and soft tissue dissection across specialties.
- #20 or #22 for longer incisions: Larger curved profiles suited to extended incisions in bigger patients or abdominal procedures.
- #11 for stab incisions: A pointed tip designed for arteriotomies, abscess drainage, or trocar site entry.
Choosing the wrong blade for the tissue and incision length creates unnecessary trauma at the wound edge. A blade that's too large for a small incision crushes tissue laterally, and a blade that's too small forces repeated passes that disrupt wound edge alignment.
Steel Quality and Edge Retention
Disposable scalpel blades are manufactured from carbon steel, stainless steel, or a combination. Carbon steel holds a sharper initial edge and cuts through tissue with less resistance. Stainless steel resists corrosion better and maintains its edge over slightly longer use.
For most single-use surgical applications, carbon steel is the preferred material because the blade is discarded after one procedure, making corrosion resistance less relevant. The priority is the sharpest possible edge at the moment of incision.
Safety Features
Safety-engineered disposable scalpels fall into three main categories.
- Retractable blade: The blade retracts into the handle after use, enclosing the sharp edge entirely.
- Shielded blade: A sliding guard covers the blade when not actively cutting, reducing exposure during hand-offs.
- Fixed blade with safety handle: The blade is non-retractable but permanently attached, removing the loading and unloading step that causes many sharps injuries with reusable handles.
An American Nurse Journal analysis of sharps data found that the OR is where almost 40% of all healthcare sharps injuries happen. Nurses pick up about 15% of those OR injuries, often from instruments they didn't use themselves. Safety-engineered designs reduce exposure for the entire surgical team, including techs and assistants who handle sharps during a case.
Cost in High-Volume Settings
Individual disposable scalpel blades cost less than a few dollars per unit. The real cost comparison is between the per-unit price of a disposable blade and the cumulative cost of reprocessing a reusable handle across hundreds of cases per year.
One Boston academic hospital ran a tray reduction project in plastic surgery during 2025. Over six months, they pulled 11,627 instruments out of the reprocessing pipeline and saved $153,089 in combined OR and sterile processing time. Disposable blades, including surgical shears, follow the same logic: fewer reusable instruments on the tray means fewer to sterilize, inspect, and reassemble.
What Happens After the Incision
A clean, atraumatic incision from a sharp disposable blade creates well-aligned wound edges that heal more predictably. A StatPearls wound healing reference notes that closure typically takes 4 to 6 weeks, and even under ideal conditions, repaired tissue tops out at about 80% of its pre-injury strength. Anything that adds trauma at the wound edge, from hypoxia to bacterial colonization, slows that recovery further.
The blade creates the incision. The closure method determines what the scar looks like. Traditional metal staples placed on the skin surface require removal in 7 to 10 days and leave characteristic railroad-track marks alongside the incision line. SubQ It! is a bioabsorbable wound closure system that places subcutaneous (under the skin) fasteners that dissolve naturally, requiring no removal visit and leaving no surface puncture marks.
For surgeons who invest in a precise incision with the right disposable blade, the closure method is where that investment either pays off or gets lost. Absorbable closure preserves the cosmetic result that a sharp, clean incision makes possible. SubQ It! SU-10 handles incisions up to 10 cm, including very small laparoscopic port sites where surface-applied devices can't operate. SubQ It! SU-25 covers incisions up to 25 cm for longer procedures.
How Does Incision Quality Affect Wound Healing and Scarring?
A sharp blade that cuts cleanly through tissue in a single pass preserves the cellular architecture on both sides of the wound edge. This allows the body's healing cascade to proceed without the extra burden of clearing damaged or crushed tissue before it can begin repair.
Several factors connect incision quality to the final scar:
- Tissue trauma at the wound edge: A dull or inappropriate blade crushes cells along the incision line, which intensifies the inflammatory phase of healing and leads to more collagen deposition, producing a wider, thicker scar.
- Healing timeline and tensile strength: Wounds generally heal in 4 to 6 weeks, and starting with minimal tissue damage gives the repair process the best chance of producing a strong, narrow scar line.
- Repeated passes with an undersized blade: Using a blade that's too small for the incision length forces the surgeon to cut across the same wound edge more than once, disrupting tissue alignment and creating irregular edges that are harder to close precisely.
- Closure method on the wound edge: Surface skin closure methods like metal staples add their own tissue trauma, puncturing the skin at regular intervals and creating the railroad-track scarring pattern. Subcutaneous closure methods place their fasteners beneath the skin surface, avoiding additional epidermal injury.
For surgeons working in plastic surgery, cosmetic procedures, or any specialty where scar appearance matters to the patient, the combination of a sharp disposable blade and a closure method that doesn't add surface trauma gives the best chance of a clean, narrow scar line.
Final Thoughts
Every disposable scalpel blade on the market delivers the same basic promise: a factory-sterile, consistently sharp, single-use cutting instrument. The differences that matter are blade geometry, safety features, and how the blade's precision connects to the rest of the surgical workflow. Choosing the right blade for the procedure and the tissue type is a clinical decision that affects incision quality, wound healing, and scarring.
Standardizing your OR tray around disposable blades that match your case mix reduces reprocessing overhead, cuts sharps injury exposure, and ensures a reliable edge for every incision. For private practices and ambulatory surgery centers running high case volumes, the operational simplification alone makes the evaluation worthwhile.
SubQ It! closes incisions from a few millimeters up to 25 cm using bioabsorbable subcutaneous fasteners that dissolve naturally, with no surface punctures, no removal visits, and no railroad-track scarring. A sharp disposable blade gives you a clean wound edge, and the right closure method preserves it. Contact us today to learn how bioabsorbable closure fits into your surgical workflow.
FAQs
1. How Often Should Disposable Scalpel Blades Be Changed During a Procedure?
Yes, many surgeons swap blades during longer cases when they feel the edge has dulled. A fresh blade ensures consistent cutting quality across tissue layers, and switching takes only a few seconds with a disposable system.
2. Can Disposable Scalpel Blades Be Used for Both Skin and Deep Tissue Incisions?
Yes, the same blade can cut through skin, subcutaneous tissue, and fascia, depending on the blade number and procedure. Blade selection should match the tissue type and depth of the planned incision.
3. What Is the Difference Between a #10 and a #15 Scalpel Blade?
The #10 has a larger curved belly suited to general skin incisions and longer cuts, while the #15 has a smaller curved profile designed for precision work in confined areas. The #15 is the more common choice for plastic surgery and small port site incisions.
4. Are Safety-Engineered Disposable Scalpels Required by OSHA?
No, OSHA doesn't mandate a specific scalpel type, but the Bloodborne Pathogens Standard requires employers to evaluate and implement safer sharps devices where clinically feasible. Safety-engineered disposable scalpels with retractable or shielded blades directly satisfy that requirement.
5. How Should Disposable Scalpel Blades Be Disposed of After Use?
Disposable scalpel blades go into a puncture-resistant sharps container immediately after use, per OSHA and facility biohazard protocols. Safety-engineered models with retractable blades can be discarded with the blade enclosed, reducing handling risk during disposal.
6. Do Disposable Scalpels Come Pre-Assembled With Handles?
Yes, fully disposable scalpels arrive with the blade integrated into a lightweight plastic handle, ready for immediate use. Some manufacturers also sell individual disposable blades designed to fit standard reusable #3 and #4 handles.


