
You've sterilized every instrument on the tray. The sterile field is set. But one dull, improperly reprocessed scalpel blade can undo all of it before you make the first incision.
Surgical site infections (SSIs) affect 0.5% to 3% of all surgical patients, and improper instrument sterilization is a known contributing factor. For the surgeon holding that blade, the difference between a clean case and a post-op complication often comes down to instrument quality at the point of use.
This post covers what makes a scalpel truly sterile, when single-use scalpels outperform reusable ones, and what to look for when choosing sterile instruments from the blade all the way through to wound closure.
Key Takeaways
- Scalpels are critical instruments that require full sterilization, not just disinfection, before every use.
- Disposable, pre-sterilized instruments deliver a consistently sharp edge and remove reprocessing variables entirely.
- The closure device you use carries the same sterility requirements as the scalpel, and choosing a single-use option keeps the sterile chain intact through the final step.
- Packaging integrity, tray optimization, and procedure-specific instrument selection all reduce contamination risk before the case even begins
- Single-use bioabsorbable closure systems remove the need for reprocessing, return visits, and staple removal, saving time for both the surgeon and the patient.
What is a Sterile Scalpel?
A sterile scalpel is a surgical blade processed to meet a sterility assurance level (SAL) of 10⁻⁶. In simple terms, that means there's no more than a one-in-a-million chance of finding a living microbe on the instrument after processing.
Infection-control guidelines classify scalpels as critical instruments because they enter sterile tissue and bone. That classification requires full sterilization after every single use. Disinfection alone isn't enough.
In practice, a sterile scalpel can be either a reusable blade that's been autoclaved between cases or a single-use, pre-sterilized disposable scalpel blade that arrives ready for immediate use. Both routes aim for the same SAL, but reliability differs depending on facility protocols and how consistent the reprocessing is.
Why Does Sterile Scalpel Selection Affect Patient Outcomes?
The blade you pick up shapes what happens at the incision site for weeks after surgery. Sterility failures and blade wear both contribute to complications that better instrument selection can prevent. Here’s why sterile scalpel selection matters:
- Infection prevention: SSIs affect surgical patients, and improper instrument sterilization is a documented outside risk factor.
- Extended hospital stays: Patients who develop SSIs after procedures like coronary bypass, bariatric, or orthopedic surgery stay 1.73 to 6.27 extra days**** compared with matched patients without infection.
- Tissue damage: Dull blades cause more physical damage at the incision site, increasing inflammation and scarring.
- Reprocessing reliability: A reusable surgical instrument has an estimated lifespan of 300 to 900 sterilization cycles, yet most facilities don't track individual cycle counts.
Each of these factors is within the surgeon's control, and the process starts with the scalpel. However, the same thinking applies beyond the blade itself. The instruments you use to close the wound, from skin closure sutures to staplers, carry the same sterility requirements.
What's the Difference Between Disposable and Reusable Sterile Scalpels?
Disposable scalpels arrive pre-sterilized and individually packaged. Reusable scalpels require cleaning, inspection, packaging, and autoclaving between every use. Both can meet sterile standards, but they carry different risk profiles.
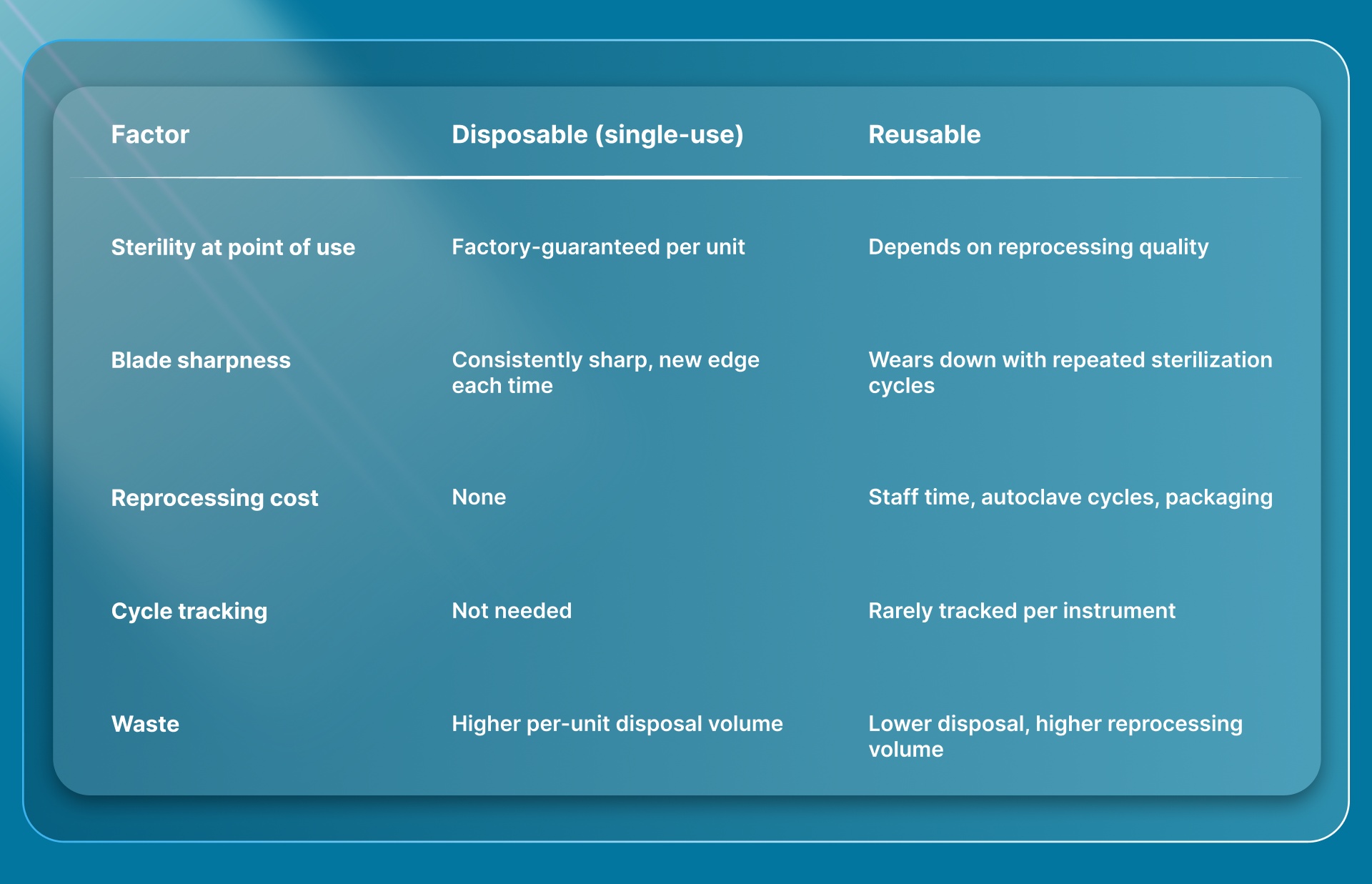
The practical trade-off comes down to how much reprocessing risk your facility can manage.
Single-use scalpels remove the reprocessing variable entirely. Every blade is sharp, verified sterile, and used once. For facilities where sterile processing department (SPD) staffing or cycle tracking is inconsistent, disposable instruments reduce one source of preventable error.
That same logic applies to other OR instruments. Disposable skin staplers and single-use wound closure devices follow the same principle of guaranteed sterility and consistent performance with no reprocessing needed.
How does Sterile Instrument Choice affect Wound Closure?
The sterile scalpel makes the incision, but the closure device finishes the job. If you're using a pre-sterilized blade to open, the instrument you choose to close should meet the same standard.
Traditional metal skin staples are typically single-use and arrive sterile. However, they sit on the skin surface, require a return visit for removal in 3 to 14 days, and leave railroad-track scarring at each puncture point. Sutures offer better cosmetic results but take significantly more time, especially for longer incisions.
Bioabsorbable closure systems take a different approach. SubQ It! places dermal fasteners under the skin. The fasteners dissolve naturally after healing is complete, so there's no removal visit, no surface puncture marks, and no railroad-track scars. Each device arrives pre-sterilized and is used once, keeping the same single-use sterility standard as a disposable scalpel.
For private practice surgeons who control their own purchasing, the closure step is where sterile instrument selection directly affects both patient outcomes and practice efficiency. A closure device that's pre-sterilized, single-use, and doesn't require a follow-up visit checks every box that matters.
What to Look for When Choosing Sterile Surgical Instruments?
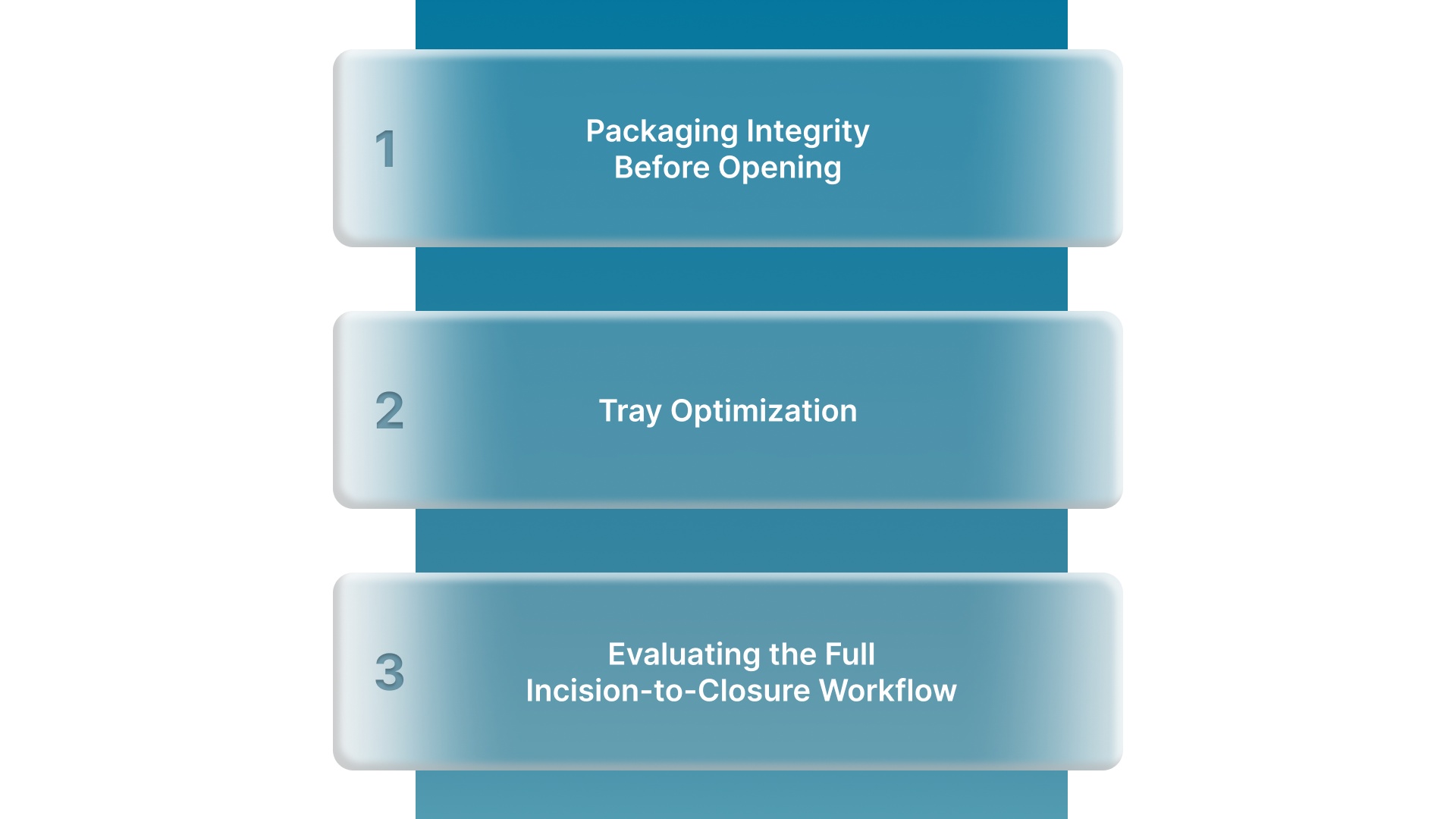
Picking the right sterile instrument starts before you enter the OR. A few practical checks at the selection and setup stage can prevent problems during the procedure.
Packaging Integrity Before Opening
Inspect every instrument package before placing it on the sterile field. Look for moisture damage, broken seals, or expired chemical indicators. If a package looks compromised in any way, don't open it. A 2025 review noted that improper packaging and storage are among the outside risk factors for surgical site infections.
For single-use instruments, factory seals make this check straightforward. For reprocessed instruments, the packaging depends on your facility's wrapping and sealing protocols, which adds another variable to monitor.
Tray Optimization
If most instruments on your tray go unused per case, your tray is too large for the procedure. Reviewing tray contents by procedure type and removing instruments that are rarely needed reduces the number of items that require reprocessing afterward. It also speeds up setup and teardown.
Some facilities build procedure-specific trays with fewer instruments and supplement with individually packaged single-use tools when needed. That approach gives you a smaller, more focused tray with less reprocessing waste.
Evaluating the Full Incision-to-Closure Workflow
It's common to focus sterile instrument decisions on the scalpel and surgical tools, but overlook the skin closure step. The closure device touches the same incision site and carries the same contamination risk. Evaluating whether your closure method is single-use, pre-sterilized, and appropriate for the incision length gives you a more complete picture of your sterile instrument workflow.
For subcuticular closure specifically, bioabsorbable options remove the need for a follow-up removal visit, which simplifies the post-op workflow for both the practice and the patient.
How do Sterile Instruments Affect OR Efficiency and Cost?
Sterile instrument management isn't just a patient safety question. For private practices and ambulatory surgical centers, it directly affects operating costs and scheduling capacity.
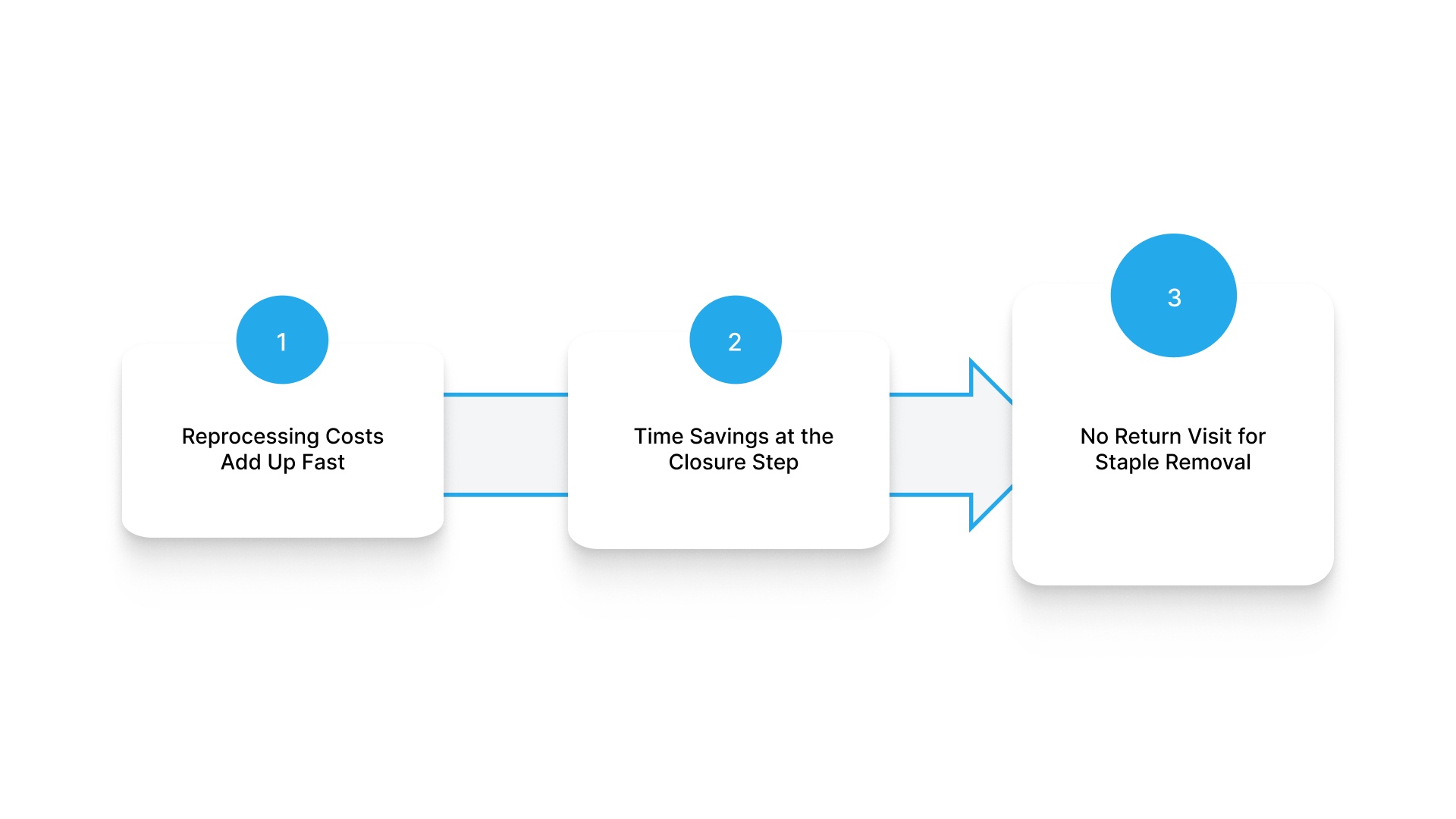
Reprocessing Costs Add Up Fast
Instrument usage rates within a standard OR tray range from only 13.0% to 21.9%. Most instruments opened for a case go unused but still require full reprocessing. One analysis estimated that a 70% decrease in unnecessary sterilization could save up to $2.8 million annually at a single center.
Every reusable instrument that goes through the autoclave costs staff time, packaging materials, and machine capacity. Selectively replacing reusable tools with single-use alternatives, where clinically appropriate, can reduce that burden without changing clinical outcomes.
Time Savings at the Closure Step
For surgeons who own their practice, time saved per case translates directly to scheduling capacity and revenue. Manual suturing of longer incisions is one of the most time-consuming steps in the OR. A skin stapler kit or bioabsorbable closure device can close the same incision significantly faster.
Faster closure means earlier room turnover, which in a private clinic setting can mean fitting one additional patient into the day's schedule. Over weeks and months, that added capacity has a real financial impact on the practice.
No Return Visit for Staple Removal
Traditional metal staples require a follow-up appointment for removal. That visit takes staff time, room availability, and patient scheduling. Bioabsorbable fasteners dissolve on their own, so there's no removal step. That's one fewer appointment for the patient and one fewer non-revenue task for the practice.
When you add up reprocessing savings, closure time savings, and the removal of follow-up visits, the cost picture shifts in favor of single-use, pre-sterilized closure devices for many procedure types.
Final Thoughts
Sterile scalpel selection is a controllable factor that directly influences SSI risk, tissue healing, scarring, and OR safety. The research points clearly toward sharper blades, verified sterility, and fewer unnecessary reprocessing cycles as practical ways to improve outcomes.
If you're evaluating your instrument protocols, start with the variables closest to the incision. Blade sharpness, sterile field timing, and closure method are three areas where small changes produce measurable results.
SubQ It! bioabsorbable dermal fasteners offer a closure method that extends that same sterile-field thinking. They're single-use, pre-sterilized, and placed under the skin with no external sharps to manage after closure. For private practice surgeons looking to save time on every case, SubQ It! SU-25 saves 7 times faster per procedure compared with manual suturing, freeing up capacity for an additional patient. Contact us today to see how it fits into your OR workflow.
FAQs
1. How Often Should Surgical Scalpels Be Sterilized?
Every scalpel blade requires full sterilization before each use on a new patient. If it's a single-use blade, it arrives pre-sterilized and is discarded after one procedure.
2. What Sterilization Methods Work for Surgical Scalpels?
Steam sterilization (autoclaving) is the most common method in the US. Ethylene oxide and gamma irradiation are also used, especially for heat-sensitive parts or factory-packaged disposable blades.
3. Can a Dull Scalpel Blade Increase Infection Risk?
Yes, dull blades cause more tissue damage at the incision site, which increases inflammation and can create conditions that favor bacterial growth. Blade condition is a recognized factor in wound healing.
4. How Long Can a Sterilized Scalpel Stay Sterile in Packaging?
The instrument stays sterile until the packaging is opened, damaged, or exposed to moisture. Storage time depends on packaging integrity, not a fixed expiration date.
5. Do Single-Use Scalpels Cost More Than Reusable Ones?
Per-unit cost is higher for disposable scalpels, but the total cost shifts when you factor in reprocessing labor, autoclave cycles, and the cost of processing unused instruments from over-packed trays.
6. What's the Difference Between Sterilization and Disinfection for Scalpels?
Sterilization removes all forms of microbial life, including bacterial spores. Disinfection lowers the microbial count but doesn't reach the spore-free standard required for instruments that contact sterile tissue.


