
Skin closure is one of the final steps in a procedure, yet it directly affects operative efficiency and the predictability of incision healing.
As a result, variability in closure method can influence operative time, follow-up requirements, and overall workflow consistency, particularly in high-volume settings.
Even small differences matter. Studies report staple closure averaging about 5.2 minutes, compared to roughly 12.6 minutes for sutures, highlighting how method selection impacts efficiency.
In this blog, you’ll examine how tissue fasteners influence surgical workflow, closure efficiency, and post-operative management, along with the role of bioabsorbable systems in modern practice.
Key Takeaways:
- Tissue fasteners are used for skin-level closure after deeper layers are secured, supporting wound approximation and primary healing.
- Different closure methods involve trade-offs between speed, consistency, and the need for follow-up care after surgery.
- Closure method selection depends on incision length, tissue characteristics, and procedural requirements.
- Tissue fasteners directly impact closure time, workflow consistency, and post-operative management across surgical cases.
- Bioabsorbable fasteners provide internal support during healing and eliminate the need for removal.
What Are Tissue Fasteners?
Tissue fasteners or wound closure devices are used to approximate and secure tissue edges during wound closure. They are most commonly at the skin level after deeper layers have been closed.
These devices function by bringing wound edges into close approximation, which:
- Supports primary intention healing by keeping tissue edges together
- Reduces the risk of wound separation (dehiscence)
- Helps minimize infection risk when proper closure is achieved
- Allows normal tissue repair processes, including epithelialization and connective tissue formation, to occur effectively
These considerations are best understood when viewed alongside real-world comparisons of commonly used closure techniques.
A Real Case Study
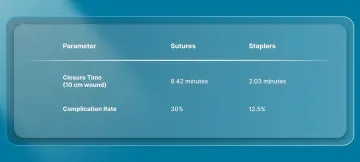
In a prospective study of 80 patients undergoing surgical procedures, wound closure outcomes using sutures and skin staples were compared to evaluate efficiency, complication rates, and overall outcomes.
- The average time required for skin closure using sutures was 8.42 minutes, compared to 2.03 minutes with skin staplers for a 10 cm wound.
- Complication rates were 30% in the suture group and 12.5% in the stapler group.
- Patient satisfaction scores showed no significant difference between the two methods.
The study also observed that the closure method influenced both procedural efficiency and post-operative outcomes, including complication rates and overall recovery experience.
These findings highlight that the choice of tissue fastener directly impacts closure time, complication risk, and workflow efficiency, reinforcing the importance of selecting appropriate closure methods based on procedural requirements.
Understanding these mechanisms clarifies how different closure methods affect workflow and postoperative management.
3 Key Types of Tissue Fasteners Used in Surgery

Tissue fasteners vary in mechanism, application method, and post-operative requirements. Selection depends on incision type, tissue characteristics, and the level of control required during closure.
Below are the key types of tissue fasteners used in the surgery.
1. Sutures
Sutures are closure devices that are used to manually approximate tissue edges. They may be absorbable or non-absorbable and are used in both deep and skin-level closure.
They provide precise control over tissue alignment and tension, making them suitable for subcuticular closure and cosmetically sensitive areas. However, they require more time and technical skill compared to mechanical closure methods.
2. Metal Skin Staples
Metal skin staples are externally applied mechanical fasteners that close incisions by bringing tissue edges together.
They are commonly used in abdominal, orthopedic, and other procedures requiring rapid closure. Staples penetrate the skin and typically require removal after initial healing, often within about 7–10 days, depending on the site.
3. Surgical Clips (Absorbable or Non-Absorbable)
Surgical clips are mechanical fasteners used primarily for vessel ligation and internal tissue approximation, especially in minimally invasive procedures.
They may be made of metal or polymer, and some variants are absorbable, while others remain permanently implanted. Their use is generally limited to internal applications rather than skin closure.
While these methods remain standard in surgical practice, newer closure systems are being developed to address the trade-offs between manual precision and mechanical speed.
For example, systems such as SubQ It! SU-25 supports skin closure across longer incisions by using a device-assisted approach that places bioabsorbable fasteners beneath the skin surface.
Although these fasteners are widely used across surgical settings, traditional closure approaches are not without practical challenges.
4 Key Limitations of Traditional Closure Methods
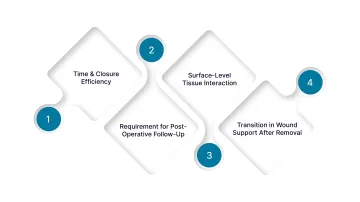
Traditional skin closure methods, primarily manual sutures and metal skin staples, remain widely used, but both present limitations that affect closure efficiency, consistency, and post-operative management.
1. Time and Closure Efficiency
Manual closure methods require more time to complete skin approximation, particularly in longer incisions or multi-case schedules.
- Closure time increases with incision length and case volume
- Small time differences per case can accumulate across operating lists
2. Requirement for Post-Operative Follow-Up
Certain closure methods require removal once initial healing is achieved, adding a step to post-operative care.
- Requires a scheduled follow-up visit for removal
- Adds administrative and clinical workload beyond the procedure
3. Surface-Level Tissue Interaction
Closure methods that rely on external skin interaction can influence the incision's behavior during early healing.
- External contact with clothing or surfaces may affect comfort
- Surface conditions, such as moisture and friction, require monitoring
4. Transition in Wound Support After Removal
Following the removal of external closure materials, the incision transitions to the tissue's intrinsic strength.
- Skin edges continue to stabilize after removal
- Temporary reinforcement may be used if additional support is required
Recognizing these constraints helps explain the rising adoption of bioabsorbable tissue fasteners in modern surgical settings.
Why Are Bioabsorbable Tissue Fasteners Gaining Adoption in Surgery?
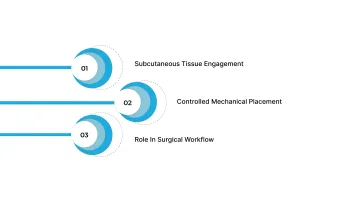
Bioabsorbable tissue fasteners represent an alternative to external, surface-based closure methods by providing internal support during healing without permanent hardware.
1. Subcutaneous Tissue Engagement
In modern closure systems, fasteners are placed within the dermal layer beneath the skin surface, allowing tissue edges to be approximated internally.
- Placement is subcutaneous, reducing the presence of external hardware on the skin surface.
2. Controlled Mechanical Placement
These systems use applicator-based devices to place fasteners at defined intervals along the incision.
- Placement depth and spacing are standardized through device design
- Device-based deployment improves consistency compared to manual suturing, though technique still influences outcomes
- Fasteners are positioned to maintain uniform approximation across the wound
3. Role in Surgical Workflow
These systems are used during the skin-closure stage after the deeper layers have been secured.
- Serve as an alternative to external skin closure methods in appropriate cases.
- Reduce the need for post-operative removal procedures
- Support procedural efficiency through device-based deployment
Alongside the reasons for their growing adoption, their practical applications across surgical specialties also deserve attention.
Where Are Bioabsorbable Tissue Fasteners Used in Surgery?
Bioabsorbable tissue fasteners are used in skin-level closure across a range of procedures where consistent approximation and simplified post-operative management are required.
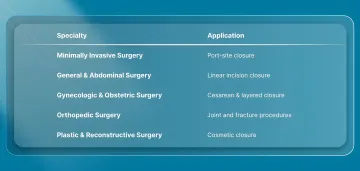
1. Minimally Invasive and Laparoscopic Procedures
Used for closing small port-site incisions created during laparoscopic and robotic procedures.
- Suitable for small, low-tension incisions such as trocar sites
- Can be applied across multiple access points in a single procedure
- Eliminate the need for removal during follow-up
2. General and Abdominal Surgery
Applied in procedures involving standard surgical incisions after deeper layers have been secured.
- Applied in procedures such as hernia repair and abdominal access incisions
- Can support consistent skin-level approximation along linear incisions
- Device-based systems may improve consistency compared to manual closure
3. Gynecologic and Obstetric Surgery
Used in procedures such as cesarean sections and gynecologic operations, where closure efficiency and wound management are important.
- Applied after the uterine and fascial layers are closed
- Maintain skin approximation during early healing
- Eliminate the need for a removal step when absorbable materials are used
4. Orthopedic Procedures
Used for skin closure following joint and bone-related surgeries.
- Applied in procedures such as joint replacement and fracture repair
- Provide an option for consistent closure along varying incision lengths
- Selection depends on wound tension, location, and procedural factors
5. Plastic and Reconstructive Surgery
Used in procedures where controlled tissue approximation is required.
- Support precise alignment in cosmetically sensitive areas
- Subcuticular approaches reduce visible external hardware
- Technique selection depends on wound type, tension, and aesthetic goals
Final Thoughts
Tissue closure decisions extend beyond the immediate procedure, influencing incision healing, recovery management, and overall case efficiency across operating schedules, particularly in longer and more complex incisions.
As procedural demands increase, closure methods that support both efficiency and predictable outcomes become increasingly important.
In this context, SubQ It! SU-25 is designed for incisions up to 25 cm and uses a device-assisted approach to place bioabsorbable fasteners beneath the skin surface.
By reducing closure time by approximately 25 minutes per case and eliminating the need for removal, it aligns skin closure with both procedural efficiency and post-operative management.
Contact us to learn how SubQ It! SU-25 fits into your surgical workflow and explores its role across surgical applications.
FAQs
1. How do bioabsorbable fasteners compare to traditional staples in terms of scarring?
Bioabsorbable subcuticular fasteners are placed beneath the skin surface, avoiding the puncture marks associated with external staples. Cosmetic outcomes are generally comparable to sutures and may improve in some cases, depending on technique, wound type, and patient factors.
2. What surgical procedures are best suited for advanced tissue fasteners?
Bioabsorbable fasteners are used in procedures such as abdominal, gynecologic, orthopedic, and reconstructive surgeries, particularly for skin-level closure. Their use depends on incision length, tissue tension, and procedural requirements rather than a single fixed indication.
3. How long do bioabsorbable fasteners provide mechanical support?
Bioabsorbable fasteners maintain strength during the early healing phase, typically the first few weeks. For example, some systems retain a significant portion of their strength for around 2–3 weeks before gradually losing support as tissue healing progresses.
4. How long does it take for bioabsorbable fasteners to be absorbed by the body?
Absorption timelines vary based on material composition. Many polymer-based fasteners degrade over several weeks to months, with some systems designed for absorption in approximately 6 to 12 weeks, while others may take longer depending on the formulation.
5. What are the cost considerations when switching to bioabsorbable fastening systems?
While device costs may be higher than traditional staples, the total cost depends on factors such as closure time, operating room efficiency, and the elimination of follow-up removal procedures. These factors can influence overall procedural and operational costs.
6. Can bioabsorbable fasteners be used for all types of wounds?
No. Their suitability depends on wound tension, location, and depth. They are generally used for low- to moderate-tension skin closures, while high-tension or complex wounds may still require traditional sutures or additional reinforcement techniques.


