Skin closure is a critical step in surgery, influencing operative efficiency, workflow consistency, and patient follow-up. In high-volume settings, even small differences in closure speed or device handling can affect throughput and coordination with sterile processing teams.
As the U.S. surgical staplers market is projected to reach USD 4 billion by 2034, the choice between reusable skin staplers and disposable systems has become increasingly important.
Metal staplers offer speed and reliable closure, while reusable systems add considerations such as reprocessing, maintenance, and consistency across repeated use.
In this blog, you’ll examine how surgeons choose the right reusable skin stapler in 2026, including cost structure, workflow integration, sterilization demands, and long-term performance.
Key Takeaways:
- Reusable skin staplers are designed for multiple uses, with sterilization and inspection between procedures to ensure consistent performance.
- Device cost depends not only on purchase price but also on reprocessing, maintenance, and workflow overhead.
- Clinical outcomes, including infection rates and wound healing, are comparable between reusable and disposable staplers when used appropriately.
- Performance can vary over time due to mechanical wear, making routine inspection and maintenance essential.
- High surgical volume and reliable sterilization workflows are critical for achieving cost-effectiveness and operational efficiency.
What Are Reusable Skin Staplers?
Reusable skin staplers are surgical devices designed for multiple patient uses after proper cleaning and sterilization between procedures. They rely on a resterilizable handle paired with single-use staple cartridges.
Most reusable skin staplers are constructed from medical-grade stainless steel, such as 316L, selected for its corrosion resistance and compatibility with repeated sterilization cycles.
From a design standpoint, reusable skin staplers typically include:

Reusable skin staplers are primarily used for rapid skin closure in linear incisions, particularly in settings where:
- Procedure volume is high
- Sterilization infrastructure is already established
- Instrument tracking and reprocessing workflows are reliable
The decision to use a reusable skin stapler is therefore less about clinical superiority and more about operational fit, reprocessing capacity, and total cost per procedure.
Once the concept of reusable skin staplers is clear, the differences from disposable systems become more meaningful.
How Reusable Skin Staplers Differ from Disposable Systems?
Choosing between a reusable skin stapler and a disposable system is less about closure mechanics and more about workflow, maintenance, and cost structure.
Both devices perform the same function intraoperatively, but how they integrate into surgical and sterilization workflows differs significantly.
1. Design and Build
A reusable skin stapler is designed for repeated use, provided it undergoes validated cleaning and sterilization between procedures. This requirement directly influences its construction and long-term performance.
- Typically built with stainless steel components for durability and corrosion resistance.
- Uses mechanical spring-driven firing systems that require periodic inspection
- May allow partial or full disassembly, depending on the manufacturer, to support cleaning
- Operates with single-use staple cartridges, while the handle is reused
2. Workflow and Sterile Processing Requirements
The primary difference between reusable and disposable systems is operational rather than clinical.

For surgical teams, the decision depends on whether the facility can support consistent, validated reprocessing without introducing delays or variability.
In settings where reprocessing capacity or turnaround time becomes a constraint, some teams evaluate alternatives that remove this dependency altogether.
Systems like SubQ It! SU-25 takes a different approach by using a single-use, subcutaneous delivery mechanism for longer incisions, eliminating the need for sterilization while maintaining procedural efficiency.
3. Clinical Performance in Practice
Reusable and disposable staplers perform similarly because they rely on the same mechanical principle of metal staple formation.
- Both provide faster skin closure compared to sutures
- Staplers deliver a consistent approximation when functioning properly
- Contaminated wounds closed with staples have a lower chance of infection than those closed with sutures.
In practice, outcomes depend more on wound selection, tissue handling, and closure technique than on whether the stapler is reusable or disposable.
4. Reliability and Device Wear
Unlike disposable systems, a reusable skin stapler introduces variability over time due to repeated use and sterilization.
- Spring fatigue can reduce firing force.
- Component misalignment may affect staple formation.
- Cartridge loading issues can develop with repeated use
- Performance depends on routine inspection, maintenance, and proper reprocessing
One of the most important differences between reusable and disposable systems is their overall cost and long-term value in clinical use.
Cost of a Reusable Skin Stapler vs Disposable Systems
The cost of a reusable skin stapler is not limited to the initial purchase. It includes reprocessing, maintenance, and workflow overhead, all of which influence the total cost per procedure compared to disposable systems.
Below are the differences between reusable skin staplers and disposable systems.
| Cost Factor | Reusable Skin Stapler | Disposable Stapler |
|---|---|---|
| Upfront cost | Higher initial investment for a reusable handle | Lower cost per unit, purchased per case |
| Per-case cost | Requires a new cartridge for each use | The entire device is used per case |
| Sterilization cost | Required between uses (reported approx. $39 per tray) | Not required |
| Maintenance | Ongoing inspection, servicing, and eventual replacement | Not required |
| Device lifecycle | Multiple uses, dependent on maintenance and reprocessing quality | Single use |
| Workflow complexity | Requires Sterile Processing and Distribution (SPD) coordination, tracking, and validation | Minimal handling, ready to use |
| Cost predictability | Variable, depends on operational efficiency | Fixed per procedure |
A closer look at how these systems differ provides context for understanding the cost drivers of reusable skin staplers.
What Drives the Cost of a Reusable Skin Stapler?
Even when the per-use cartridge cost is lower, the overall cost depends on how efficiently the system is managed.
- Additional overhead includes cleaning and decontamination, inspection and quality assurance, sterilization validation, and packaging.
- Device tracking and coordination between the OR and sterile processing add operational complexity.
As a result, cost efficiency depends more on workflow performance and reprocessing consistency than on the device itself.
Once the cost components are clear, it becomes easier to assess when the investment in a reusable system is justified.
When a Reusable Skin Stapler Becomes Cost-Effective?
A reusable skin stapler may be cost-effective under specific conditions:
- Procedure volume is consistently high
- Sterile processing workflows are well-established and reliable
- Devices are used frequently enough to distribute capital and operational costs
In lower-volume settings or where sterilization workflows are inconsistent, reprocessing overhead can offset potential savings.
Once cost-effectiveness is clear, it becomes important to consider how newer bioabsorbable systems influence traditional cost structures.
How Bioabsorbable Systems Change the Cost Model?
Bioabsorbable closure systems introduce a different cost structure by removing both reprocessing and removal steps.
- Single-use, pre-sterilized devices eliminate the need for sterilization workflows.
- No removal step reduces post-procedure resource use
- Workflow becomes more predictable, with cost tied directly to each procedure rather than operational variability
This shifts evaluation beyond reusable versus disposable alone, toward overall procedural efficiency and follow-up resource use.
Alongside cost evaluation, attention must also be given to the sterilization and upkeep required for reusable devices.
4 Key Sterilization & Maintenance Requirements for Reusable Skin Staplers
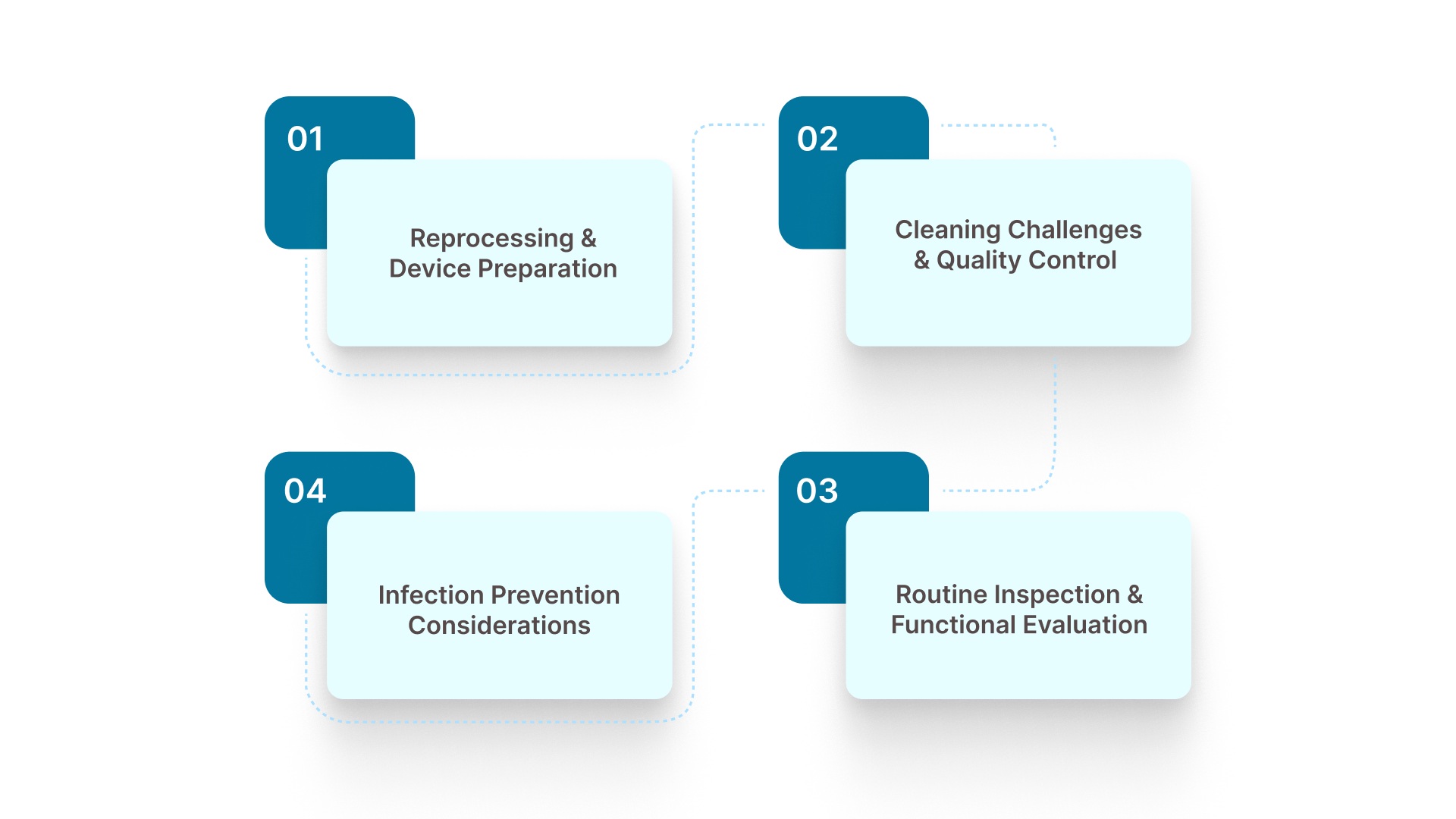
Reusable skin staplers must be cleaned and sterilized between uses so they remain safe and effective for multiple procedures.
Proper reprocessing includes thorough cleaning, inspection, and sterilization to prevent residual biological material and reduce the risk of contamination.
1. Reprocessing and Device Preparation
Reusable medical devices require a multistep process to prepare them for subsequent use, which typically includes:
- Disassembly of removable components when indicated by the manufacturer
- Manual pre‑cleaning to remove visible debris immediately after use
- Automated washing in a validated washer-disinfector or equivalent
- Inspection for residue, wear, or damage after cleaning
- Packaging with appropriate indicators prior to sterilization
- Terminal sterilization (for example, steam sterilization under pressure)
- Documentation of cleaning and sterilization cycles
Thorough cleaning is important because inadequately cleaned instruments can retain biological debris that may compromise sterilization effectiveness.
2. Cleaning Challenges and Quality Control
Reusable skin staplers may include internal mechanisms such as springs and hinges that can trap debris if not properly accessed during cleaning. Incomplete cleaning may increase the risk of persistent microbial contamination.
Effective reprocessing relies on validated procedures and consistent execution. Many facilities use chemical and sterilization indicators to confirm that sterilization conditions are met during each cycle.
3. Routine Inspection and Functional Evaluation
Routine checks before sterilization help ensure that a reusable skin stapler remains ready for use. Common inspection points include:
- Mechanical wear or visible damage on external surfaces
- Smooth handling and firing mechanics
- Proper engagement of cartridges with the stapler body
Mechanical issues that may arise over time include:
- Misalignment or difficulty firing due to wear
- Spring tension loss that reduces firing consistency
- Debris buildup that affects moving parts
If a device does not function reliably or shows signs of wear that could affect performance, it should be retired from use.
4. Infection Prevention Considerations
Cleaning and sterilization efforts are intended to eliminate contamination and reduce the risk of surgical site infection. Devices that penetrate or contact non‑intact skin require sterilization to destroy all microorganisms present on the instrument surface.
Persistent biological films can resist sterilization if not removed during earlier cleaning steps. Ensuring all reprocessing steps are completed effectively helps safeguard patient safety.
Once sterilization and maintenance requirements are understood, it becomes easier to evaluate which system best meets clinical needs.
How to Choose the Right Reusable Skin Stapler System in 2026?
Selecting a reusable skin stapler affects workflow, patient experience, and procedural efficiency. The right choice depends on how well the system fits your facility and surgical volume.
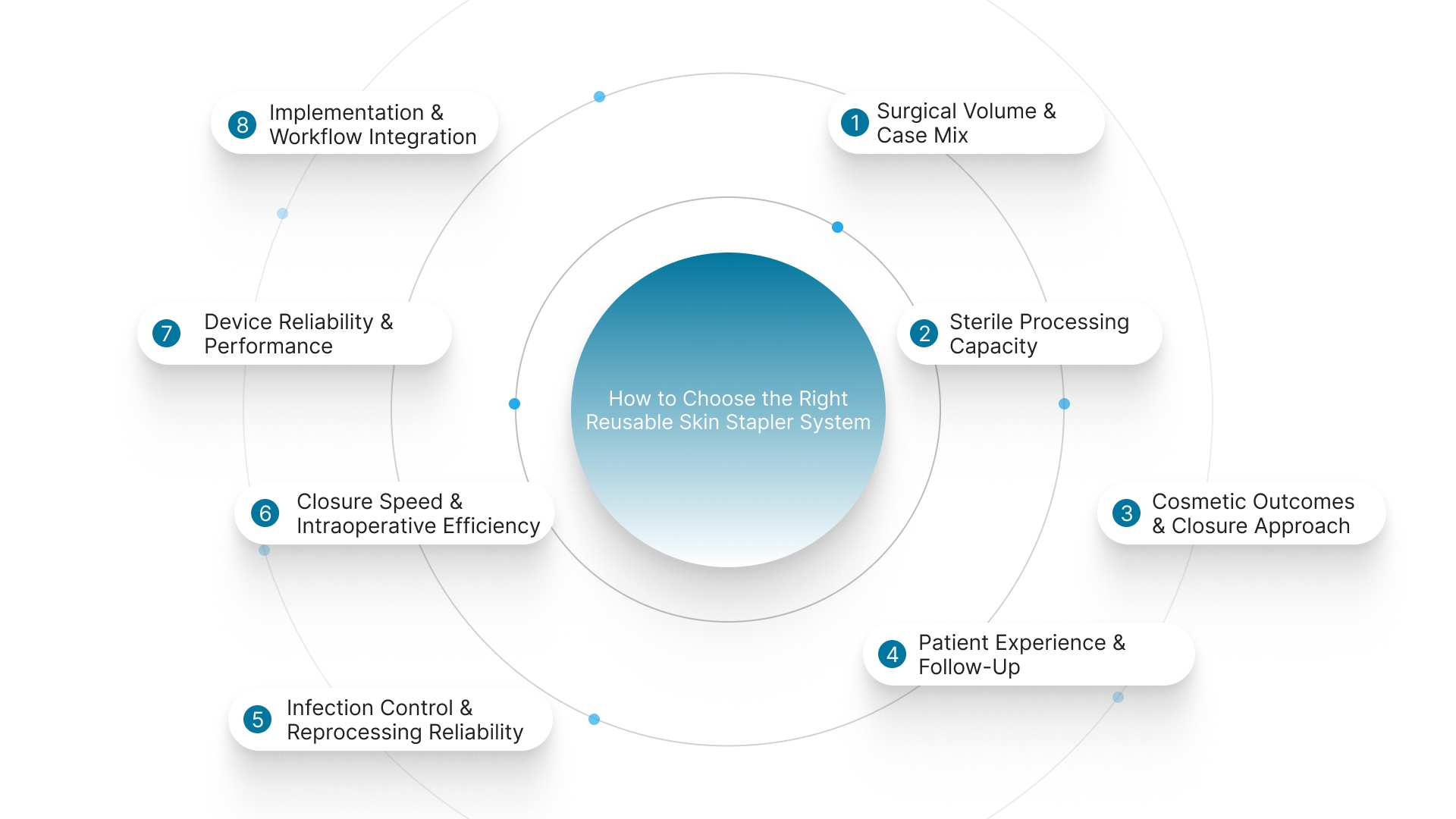
1. Surgical Volume and Case Mix
High-volume facilities benefit most from reusable skin staplers because frequent use distributes the device’s cost and justifies reprocessing efforts. Lower-volume settings may find the overhead less efficient.
- Ideal for high-volume procedures where repeated use is expected
- Suitable for routine closures in inpatient or high-throughput environments
2. Sterile Processing Capacity
Reusable staplers require reliable cleaning and sterilization between uses. Workflow integration with sterile processing is crucial for device performance and patient safety.
- Requires access to sterilization equipment and trained staff
- Consistent cleaning, inspection, and sterilization protocols are essential
- Coordination between the OR and sterile processing teams is necessary
3. Cosmetic Outcomes and Closure Approach
The type of stapler affects the appearance of the wound after healing. Traditional metal staplers, even reusable ones, can leave small puncture marks, whereas subcutaneous or bioabsorbable systems minimize surface scarring.
- Metal staplers can leave visible puncture marks or “train track” scarring, especially in cosmetically sensitive areas.
- Bioabsorbable systems provide subcuticular closure with minimal surface marks.
4. Patient Experience and Follow-Up
The patient experience is affected by whether a device requires removal after healing. Follow-up visits increase patient burden and clinic workload.
- Metal staplers require a follow-up removal visit.
- Bioabsorbable fasteners are absorbed naturally, eliminating the need for removal visits.
5. Infection Control and Reprocessing Reliability
Infection risk is tied to cleaning quality and device management. Proper reprocessing ensures safe use of reusable staplers, while single-use devices remove variability.
- Reusable devices must be cleaned, inspected, and sterilized properly
- Single-use systems provide consistent sterility per case
- Infection outcomes depend largely on adherence to validated protocols
6. Closure Speed and Intraoperative Efficiency
All stapler systems close skin faster than manual suturing, reducing operative time. Some subcutaneous fastener systems can further improve efficiency.
- Faster closure improves throughput in high-volume or lengthy procedures
- Device choice can affect total procedural time, especially for repetitive closures
7. Device Reliability and Performance
Long-term reliability is important for reusable staplers. Devices should be evaluated for consistent firing force and staple formation across repeated uses.
- Inspect for mechanical wear, misalignment, or cartridge issues
- Performance depends on consistent maintenance and reprocessing
8. Implementation and Workflow Integration
Adopting a reusable stapler requires structured staff training and workflow alignment to maintain efficiency and safety.
- Train clinical staff on handling and closure techniques
- Ensure sterile processing staff understand cleaning, inspection, and documentation
- Integrate device tracking, inventory management, and maintenance schedules
Final Thoughts
Effective skin closure shapes operative efficiency, postoperative care, and consistent outcomes across surgical cases.
As surgical teams prioritize speed, reliability, and predictability, advanced closure systems become increasingly valuable in managing workflow and patient experience.
In this context, SubQ It! SU-25 is designed for longer incisions, supporting closure of up to 25 cm while reducing closure time by approximately 25 minutes per case.
This can be particularly relevant in high-volume or private practice settings where closure time affects throughput and scheduling.
Contact us today to explore how SubQ It! SU-25 fits into your surgical workflow and supports more efficient, consistent closure.
FAQs
1. How does a reusable skin stapler differ from a disposable skin stapler?
A reusable skin stapler is sterilized and reused across procedures with new cartridges, while a disposable stapler is pre-sterilized and discarded after a single use.
2. What are the typical clinical indications for a reusable skin stapler?
A reusable skin stapler is used to rapidly close linear, low-tension incisions, especially in general surgery, trauma, and high-volume settings.
3. Can reusable skin staplers still use single-use cartridges?
Yes, reusable skin staplers still use single-use cartridges. This ensures sterility and consistent closure performance while allowing the main stapler mechanism to be safely reprocessed between procedures.
4. Are there limitations to where a reusable skin stapler can be applied?
Reusable skin staplers are ideal for linear, low-tension wounds, but they are less suitable for:
- Areas with high movement, like joints
- Wounds that are irregular or jagged
- Cosmetically sensitive regions, such as the face or hands
- Deep or multilayer wounds where additional closure methods (sutures, adhesives, subcuticular fasteners) are needed
5. What factors should influence facility decisions about adopting a reusable skin stapler?
Facility decisions should consider:
- Sterilization workflow and reprocessing capacity
- Surgical volume to ensure repeated use
- Cost structure compared to disposables or bioabsorbable systems
- Staff training for proper device handling and inspection
- Clinical priorities, including closure speed, cosmetic outcomes, and patient experience
6. Are there surgical situations where a reusable skin stapler should not be used?
Yes. Even with a reusable system, staplers may not be optimal for:
- High-tension wounds where edges cannot approximate easily
- Irregular or jagged wound shapes
- Cosmetically sensitive areas requiring precise closure