Fascial closure may take only a few minutes, yet it determines whether the port site maintains its integrity afterward. Although overall hernia rates remain low, the risk increases with larger ports, midline access, and repeated manipulation.
In laparoscopic settings, closure is performed through limited access, where depth and tissue capture are not always consistent. To address this challenge, fascial closure devices support controlled suture placement and improve consistency in confined spaces.
Their growing role is reflected in market projections, with the global fascial closure system market expected to reach USD 85.2 million by 2032.
In this blog, you’ll explore how fascial closure devices work, what sets them apart, and how to select the most appropriate option based on procedural requirements.
Key Takeaways:
- Fascial closure is a risk-based decision driven by port size, location, and intraoperative factors, including manipulation and defect enlargement.
- Ports ≥10 mm and midline or umbilical sites carry a higher hernia risk and typically require reliable, full-thickness closure.
- Device selection depends on procedural fit, tissue access, and the ability to maintain consistent suture placement under limited access conditions.
- Closure effectiveness is influenced by technique and execution, with risks such as incomplete capture or improper placement leading to failure.
- Different surgical scenarios, including bariatric cases and extraction sites, require adjustments in the closure approach to maintain stability and reduce complications.
What Is a Fascial Closure Device?
A fascial closure device is a surgical instrument used to close the fascial layer after laparoscopic or open procedures, especially at trocar or port sites.
These devices are specifically designed to facilitate secure closure of abdominal fascia, which is critical in preventing port-site complications such as hernias.
Here are the key functions of a fascial closure device:

Before examining the situations where it becomes necessary, it is important to understand what a fascial closure device is and its role during closure.
Situations where closure decisions may vary:
- Ports <10 mm: Often do not require closure unless risk factors are present
- Low-risk cases: Small, non-midline ports with minimal manipulation may not require formal closure
This makes fascial closure a risk-based decision, where device use becomes important when consistent, full-thickness closure is difficult to achieve manually.
In cases involving smaller port sites or minimally invasive access, once fascial closure is not required or has been addressed, attention shifts to the skin layer. For these smaller incisions, systems like SubQ It! SU-10 provides a bioabsorbable subcutaneous closure option that avoids external staples and their removal.
The specific clinical need often influences the type of device selected, making it important to understand how available options differ in design and application.
4 Key Types of Fascial Closure Devices and How They Differ
Fascial closure devices differ based on how they enable suture placement, guide tissue access, and control closure consistency during fascial repair.
These differences directly influence how reliably fascia is approximated, particularly in laparoscopic procedures where access, depth, and visualization are constrained.
Below are the device types, defined by their mechanisms and how they function in practice.
1. Suture Passer Devices
Suture passer devices use a needle-based mechanism to pass a suture through the fascial layer, typically via percutaneous insertion near the port site. They are widely used because they allow flexibility in technique and material selection.
Key characteristics include:
- Needle-driven suture passage: Passes suture through fascia using a sharp or blunt needle, enabling full-thickness closure when aligned correctly across both tissue edges
- Manual control over placement: Allows adjustment of needle angle and depth to match tissue thickness and port position during closure
- Flexible suture selection: Supports use of different suture materials based on strength requirements, tissue quality, and surgeon preference
- Broad procedural use: Applicable across a wide range of port sizes and commonly used in standard laparoscopic workflows
2. Guided Suture Systems
Guided systems incorporate built-in guides or channels that direct needle placement through the fascia. These systems are designed to standardize where the suture enters and exits the tissue.
Key characteristics include:
- Controlled needle trajectory: Guides the position of the needle at a fixed distance from the fascial edge, helping maintain consistent tissue bite placement
- Improved placement consistency: Reduces variation in suture entry and exit points compared to freehand techniques in limited access conditions
- Structured tissue access: Helps maintain alignment of the device with the fascial defect, especially when working through trocar-based entry points
- Designed for laparoscopic access: Integrates with port-based workflows, supporting closure without extensive repositioning during the procedure
3. Mechanical Closure Systems
Mechanical systems use preloaded components such as anchors or deployment mechanisms to approximate fascia without traditional needle passing. These devices are designed to simplify closure steps and standardize approximation.
Key characteristics include:
- Predefined deployment mechanism: Uses a controlled trigger-based system to deliver closure components, reducing dependence on manual suture passing technique
- Reduced manual handling: Minimizes repeated needle passes by using a single-step or limited-step deployment process
- Consistent approximation pattern: Delivers a uniform closure configuration across the defect, improving repeatability across cases
- Self-contained design: Typically preloaded with closure components, reducing the need for additional suture preparation during the procedure
4. Integrated Trocar-Based Systems
Integrated systems combine trocar access and closure functionality, allowing suture placement through the existing port without removing it. This supports closure under maintained visualization.
Key characteristics include:
- Closure through trocar access: Enables suture placement via the existing cannula, avoiding the need for additional percutaneous entry points
- Maintains pneumoperitoneum: Allows continuous visualization of internal structures during closure without loss of insufflation
- Simplified workflow: Reduces procedural steps by combining access and closure within the same system
- Aligned with port dimensions: Designed to match standard trocar sizes, supporting consistent placement relative to the fascial defect
Once you understand how these devices differ in design, it also clarifies how they function during actual surgical use.
How Fascial Closure Devices Work in Practice?
Fascial closure devices are used at the end of a procedure to facilitate suture placement across the fascial defect, typically at trocar or port sites.
Their role is to support consistent, full-thickness fascial approximation, particularly in settings with limited access and where closure must be performed through small entry points.
Below are the steps outlining how these devices are typically used during closure.
1. Device Positioning at the Port Site
The device is introduced either percutaneously adjacent to the port or through the existing trocar, depending on the system being used. Accurate positioning is required to align the device with the fascial defect before suture passage.
Key considerations include:
- Alignment with fascial edges: Positioning must target both sides of the defect to allow proper suture placement across the fascia
- Access approach: Device insertion may be external or through the cannula, based on the closure system design
- Stability during insertion: Maintaining steady positioning helps ensure controlled engagement of the tissue layers
2. Tissue Penetration and Suture Passage
The device is advanced through the abdominal wall to penetrate the fascia and deliver or retrieve suture. This step plays a critical role in achieving full-thickness closure across the defect.
Key considerations include:
- Depth control: The device must pass through the fascial layer without remaining too superficial or extending beyond the intended tissue plane
- Suture delivery or retrieval: Depending on the mechanism, the device either places or captures the suture across the defect
- Repeat passage: The process is typically performed on both sides of the defect to complete one suture placement cycle
3. Suture Placement Across the Defect
Once both sides of the fascia are engaged, the suture is positioned across the defect to approximate the tissue edges. This establishes the structural basis for closure.
Key considerations include:
- Symmetric tissue capture: Both fascial edges must be engaged evenly to avoid gaps or uneven tension
- Adequate bite size: Tissue bites should be sufficient to prevent suture pull-through under physiological stress
- Consistent placement pattern: Multiple sutures may be placed in a uniform pattern along the defect for larger port sites
4. Approximation and Securing of Fascia
The suture is then tensioned and secured, bringing the fascial edges together to complete the closure. The integrity of closure depends on both placement and controlled tensioning.
Key considerations include:
- Controlled tension: Fascia should be approximated without excessive tightening that may compromise tissue perfusion
- Closure confirmation: Visual or tactile assessment is used to confirm proper approximation of the fascial edges
- Sequential closure: Larger defects may require multiple sutures placed along the incision for complete closure
5. Final Inspection Before Port Removal
Before removing the port, the closure is assessed to confirm that the fascial defect has been fully approximated and no gaps remain.
Key considerations include:
- Internal visualization (when maintained): Allows confirmation of closure from within the abdominal cavity
- Edge alignment check: Ensures the fascial edges are properly approximated without overlap or separation
- Defect coverage: Confirms that the entire port site has been adequately closed before completing the procedure
The way these devices function in practice shapes the criteria for determining which option is most appropriate for a given case.
How to Choose the Right Fascial Closure Device?
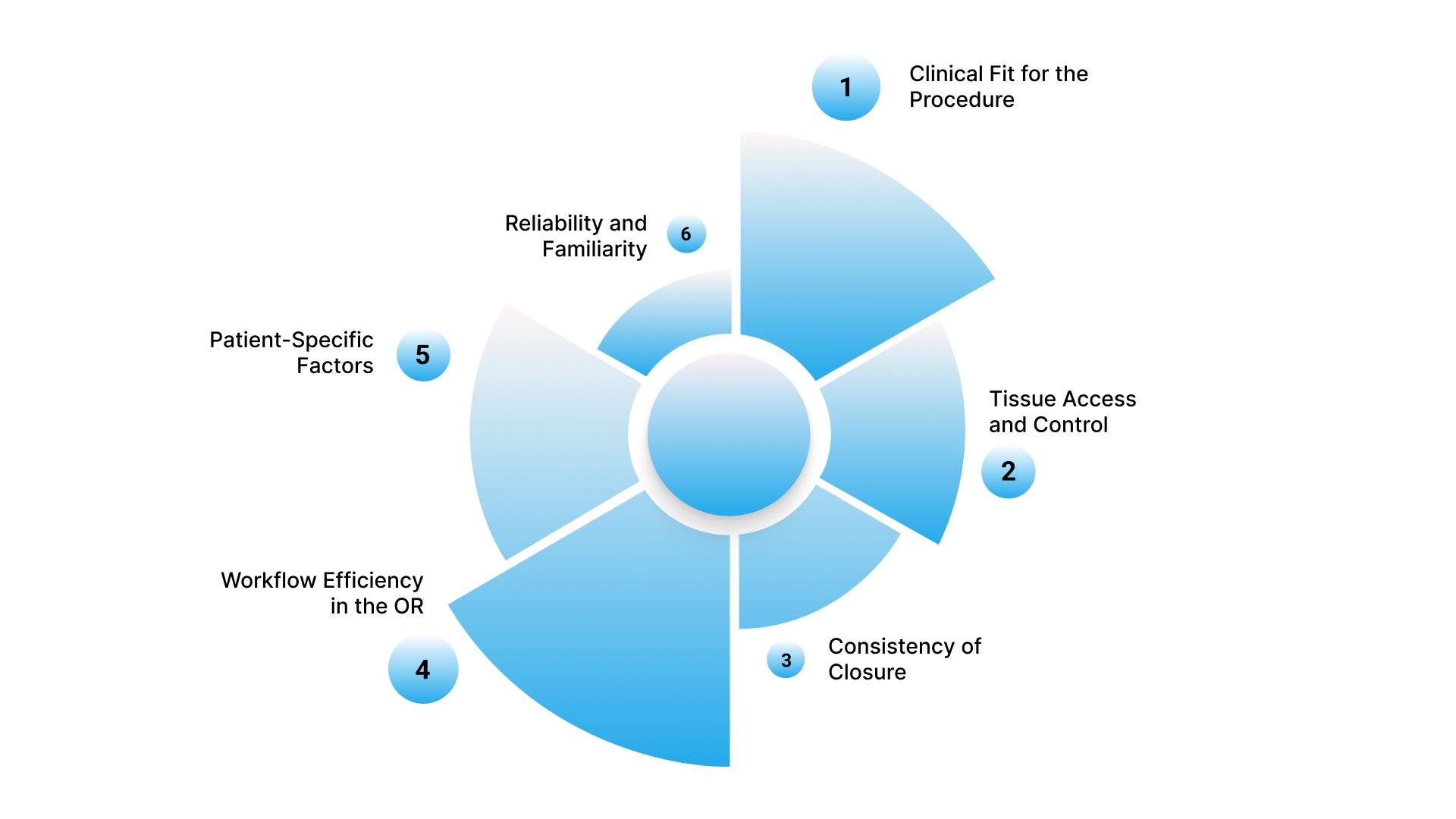
Choosing a fascial closure device depends on how well it supports consistent, controlled closure under specific procedural conditions.
Selection is guided by factors such as port characteristics, tissue access, and the device's reliability within the constraints of the procedure.
Here’s how you can choose the right fascial closure device:
1. Clinical Fit for the Procedure
The device must align with the type of procedure and characteristics of the fascial defect, including port size, location, and extent of manipulation during the case.
Key considerations include:
- Port size compatibility: Device should support closure of ≥10 mm ports and larger defects created during specimen extraction
- Defect location: Midline and umbilical sites require more reliable closure due to higher hernia risk
- Extent of manipulation: Ports subjected to repeated instrument movement may have enlarged defects requiring controlled closure
2. Tissue Access and Control
Device selection depends on how effectively it allows controlled access to the fascial layer across varying patient and procedural conditions.
Key considerations include:
- Depth handling: The device should allow consistent engagement of the fascia across different abdominal wall thicknesses
- Placement precision: Enables accurate suture placement across both sides of the defect
- Stability during use: Maintains alignment during insertion and suture passage
3. Consistency of Closure
The device should support repeatable closure performance, helping maintain uniform technique across cases.
Key considerations include:
- Reproducibility: Supports consistent suture placement across multiple uses
- Reliable tissue capture: Enables engagement of both fascial edges when used correctly
- Uniform placement pattern: Helps maintain consistent spacing in larger defects
4. Workflow Efficiency in the OR
The device should integrate into the existing procedural workflow without adding unnecessary complexity.
Key considerations include:
- Number of steps required: Fewer steps can reduce variability during closure
- Compatibility with setup: Should work with existing ports and instruments
- Ease of handling: Should not disrupt the pace of closure
5. Patient-Specific Factors
Patient characteristics influence how easily the fascia can be accessed and closed.
Key considerations include:
- Abdominal wall thickness: Increased thickness may require devices that support deeper access
- Tissue quality: Fragile or previously operated tissue may require more controlled placement
- Defect size variability: Larger or irregular defects may require flexible suture placement
6. Reliability and Familiarity
Device performance and familiarity influence how consistently closure can be performed.
Key considerations include:
- Predictable performance: Device should function consistently during repeated use
- Learning curve: Ease of adoption based on the mechanism and handling
- Team coordination: Should align with the assistant and staff workflow during closure
The selection process becomes more practical when examined across different surgical scenarios, where procedural demands and patient factors vary.
Fascial Closure Device Considerations Across Surgical Scenarios
Fascial closure requirements vary based on how the defect is created, where it is located, and the mechanical stress it will experience during healing.
Device use should be evaluated within these conditions, as closure demands differ across procedural scenarios.
- Laparoscopic multi-port procedures: Closure is performed at selected port sites, with requirements determined by port size, location, and the constraints of trocar-based access
- Single-port and reduced-port surgery: A single access site creates a larger fascial defect with concentrated stress, requiring consistent approximation across the entire opening
- Bariatric and high BMI cases: Increased abdominal wall thickness and intra-abdominal pressure require reliable depth control and consistent tissue engagement during closure
- Procedures involving specimen extraction: Enlarged or extended defects require multiple, evenly spaced sutures to distribute tension and achieve complete fascial approximation
- Reoperative or compromised tissue cases: Altered tissue planes and reduced fascial strength require precise suture placement to maintain stable closure across the defect
These scenario-based considerations also highlight where complications can arise, making it important to examine potential risks and common failure points.
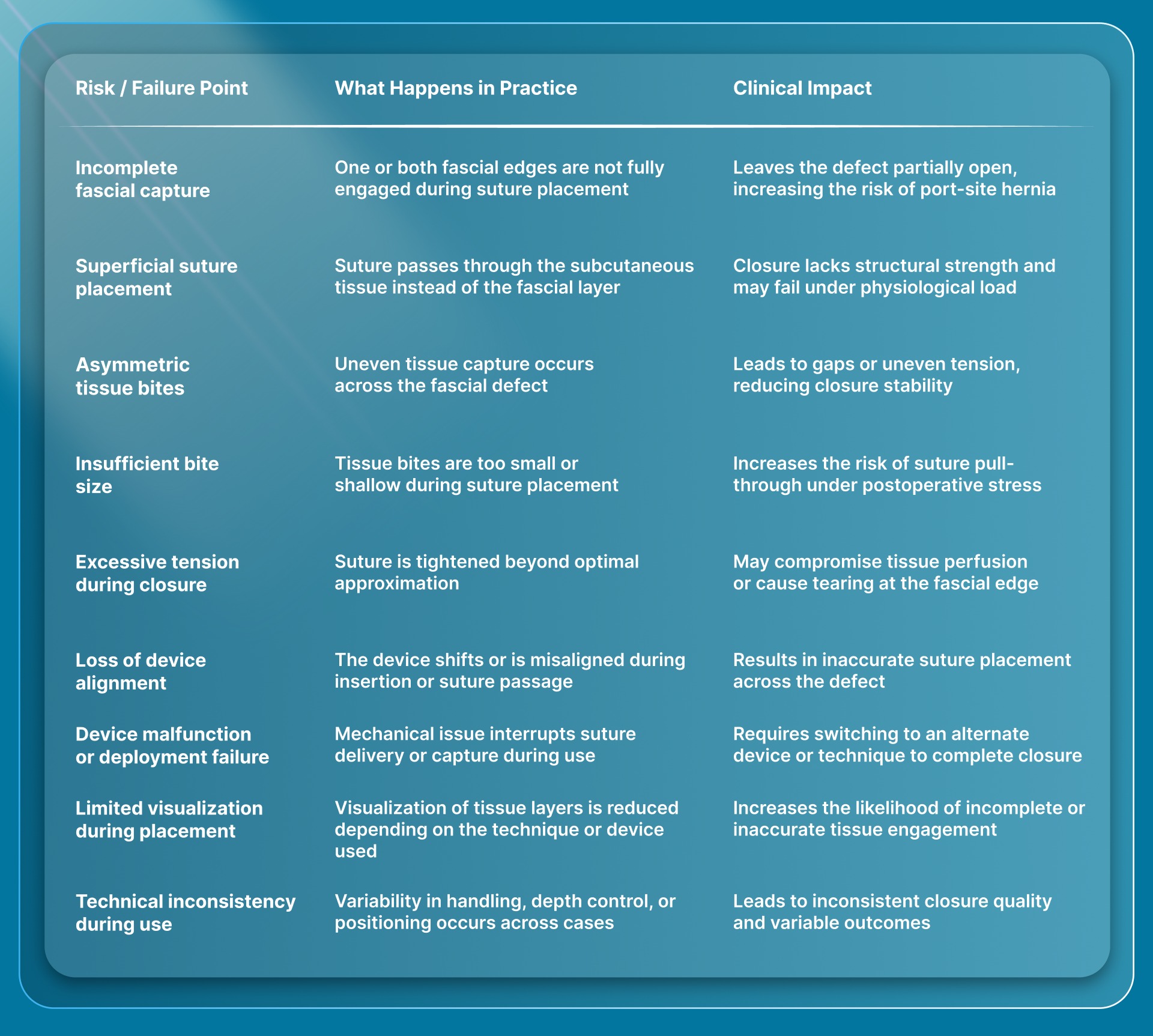
Risks and Failure Points With Fascial Closure Devices
Fascial closure reliability depends on accurate tissue capture, proper suture placement, and controlled execution under intraoperative conditions. Below is a table outlining common failure points, what happens in practice, and their clinical implications.
Final Thoughts
Choosing the right fascial closure approach directly affects how reliably the port site holds after surgery. As procedures rely on smaller access points, consistency during closure becomes more important.
Once fascial closure is complete, attention shifts to the skin layer, where closure technique directly affects follow-up requirements and visible outcomes.
In this context, SubQ It! SU-10 offers a bioabsorbable, subcutaneous skin closure system for smaller surgical incisions, placing 10 fasteners beneath the skin to close incisions up to 10 cm with precise edge approximation.
Because the fasteners are bioabsorbable and placed under the skin, there is no need for a return visit for staple removal, and they also support more efficient closure in laparoscopic workflows.
Contact us to explore how SubQ It! SU-10 can support post-biopsy healing and cosmetic outcomes, and learn more about this advanced closure solution.
FAQs
1. Do fascial closure devices reduce operative time compared to manual closure?
In selected cases, fascial closure devices can reduce closure time by simplifying suture placement, particularly in limited access settings. The time benefit depends on device design, familiarity, and defect complexity.
2. Can fascial closure devices be used in pediatric cases?
Yes, but device selection must account for smaller port sizes and thinner abdominal walls. Careful depth control and appropriate sizing are essential to ensure accurate fascial engagement.
3. How does pneumoperitoneum affect fascial closure device use?
Maintaining pneumoperitoneum during closure preserves internal visualization, helping confirm accurate needle passage and suture placement. It also reduces the risk of unintended tissue capture.
4. Are fascial closure devices compatible with all trocar systems?
No, compatibility varies by port size, access method, and device design. It should be confirmed before use to ensure proper alignment and consistent suture placement.
5. What role does assistant support play during device-assisted closure?
Assistant coordination helps maintain alignment, stabilize insertion, and manage suture handling, supporting more consistent closure, especially in technically demanding cases.
6. Can fascial closure devices be used in emergency procedures?
Yes, but selection depends on availability and familiarity. In time-sensitive situations, simpler systems with fewer steps are often preferred for reliable execution.