
One mesh brand rarely fits every hernia on your schedule. An inguinal repair in a healthy 35-year-old and a contaminated ventral hernia in a patient with prior mesh failure require completely different materials. Defaulting to a single product across both cases creates avoidable risk.
Roughly 27% of men will develop an inguinal hernia in their lifetime, which makes mesh one of the highest-volume purchasing decisions in general surgery. Getting it right means matching the brand and material to the clinical scenario.
Here's a 2026 breakdown of hernia mesh brands by material type, with practical criteria for matching mesh to hernia.
Key Takeaways
- Mesh selection is one of the highest-volume purchasing decisions in general surgery, with US hernia repairs exceeding 1 million per year.
- Hernia mesh falls into four material categories: synthetic polypropylene, composite/barrier, biologic, and bioabsorbable synthetic, each suited to specific clinical scenarios.
- Biologic mesh carried a 24.6% ventral hernia recurrence rate in a 2025 pooled analysis, while synthetic mesh recurred at 9.7%.
- In North America, roughly 6.04% of inguinal hernia patients develop chronic postoperative pain, a rate that varies with mesh type, fixation method, and nerve handling.
- Ventral hernia repair with biologic mesh ran $80,420 per patient on average, compared to $61,036 with synthetic, a gap driven by prosthetic cost and longer OR time.
What Are the Main Types of Hernia Mesh?
Hernia mesh products fall into four material categories, each with distinct properties that influence where and how they're used. Here's how they compare at a glance.
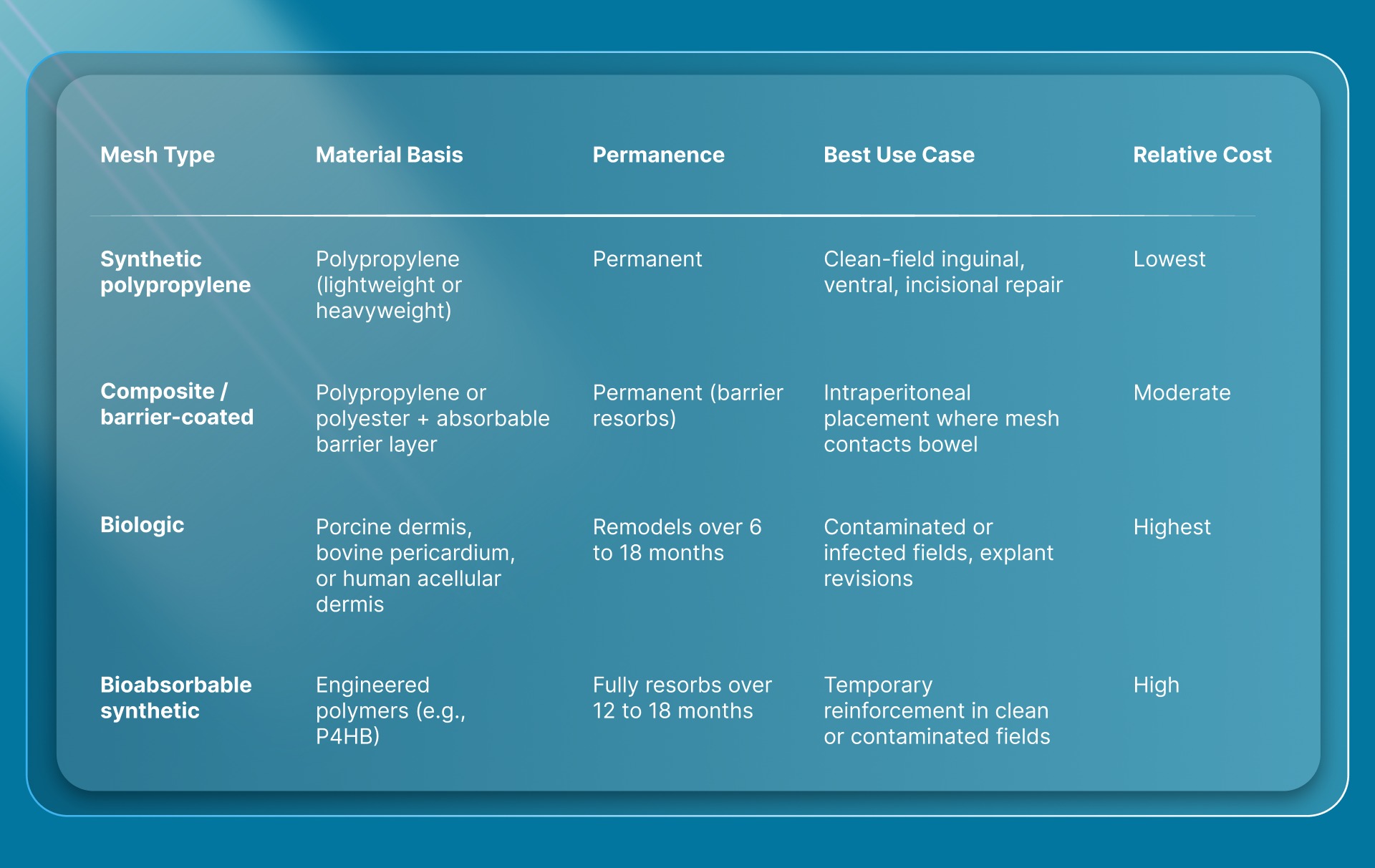
Knowing these categories makes it easier to evaluate the brand names within them.
Synthetic Polypropylene Mesh
Synthetic polypropylene remains the most widely implanted hernia mesh material. It provides permanent tensile strength, resists infection, and integrates with native tissue through fibrous ingrowth. Lightweight and macroporous versions have largely replaced heavyweight designs, reducing foreign body sensation and chronic stiffness.
Most inguinal, ventral, and incisional hernia repairs in clean surgical fields use a synthetic polypropylene product. The material is inexpensive relative to biologics and carries the longest published track record for recurrence outcomes.
Composite and Barrier-Coated Mesh
Composite meshes combine a permanent structural layer (typically polypropylene or polyester) with a barrier coating on one or both surfaces. The barrier, often made from oxidized regenerated cellulose, collagen, or hydrogel, prevents adhesion formation when mesh contacts visceral organs during intraperitoneal placement.
These products are used primarily in laparoscopic ventral hernia repair and any open case where the mesh will be positioned against bowel. The barrier layer resorbs over weeks, leaving the permanent mesh in place. Abdominal wound closure in these cases often involves both fascial and skin layers, adding time to the procedure.
Biologic Mesh
Biologic meshes are derived from human or animal tissue (typically porcine dermis, bovine pericardium, or human acellular dermis). They're designed to be remodeled by the patient's own collagen over time rather than remaining as a permanent implant. Biologic meshes are significantly more expensive per unit than synthetics.
When matched against biologic repairs in contaminated hernia cases, synthetic sublay mesh held up better on both wound complications and durability (2024 data).
Biologics still have a role in heavily contaminated or infected fields where permanent synthetic material carries higher explantation risk. The decision depends on contamination grade, patient comorbidities, and cost tolerance.
Bioabsorbable Synthetic Mesh
Bioabsorbable synthetic meshes are fully resorbable implants made from engineered polymers. They provide temporary scaffold support while native tissue regenerates, then dissolve completely. These products occupy a niche between permanent synthetic mesh and biologic options.
The most established bioabsorbable mesh uses poly-4-hydroxybutyrate (P4HB). This category is newer than the others, and long-term recurrence data beyond five years is still accumulating. Absorbable closure devices apply a similar bioabsorbable principle to the skin closure step of surgery.
Which Hernia Mesh Brands Are Surgeons Using in 2026?
Five manufacturers account for the majority of hernia mesh products in US operating rooms. Here's a quick overview and side-by-side comparison of them:
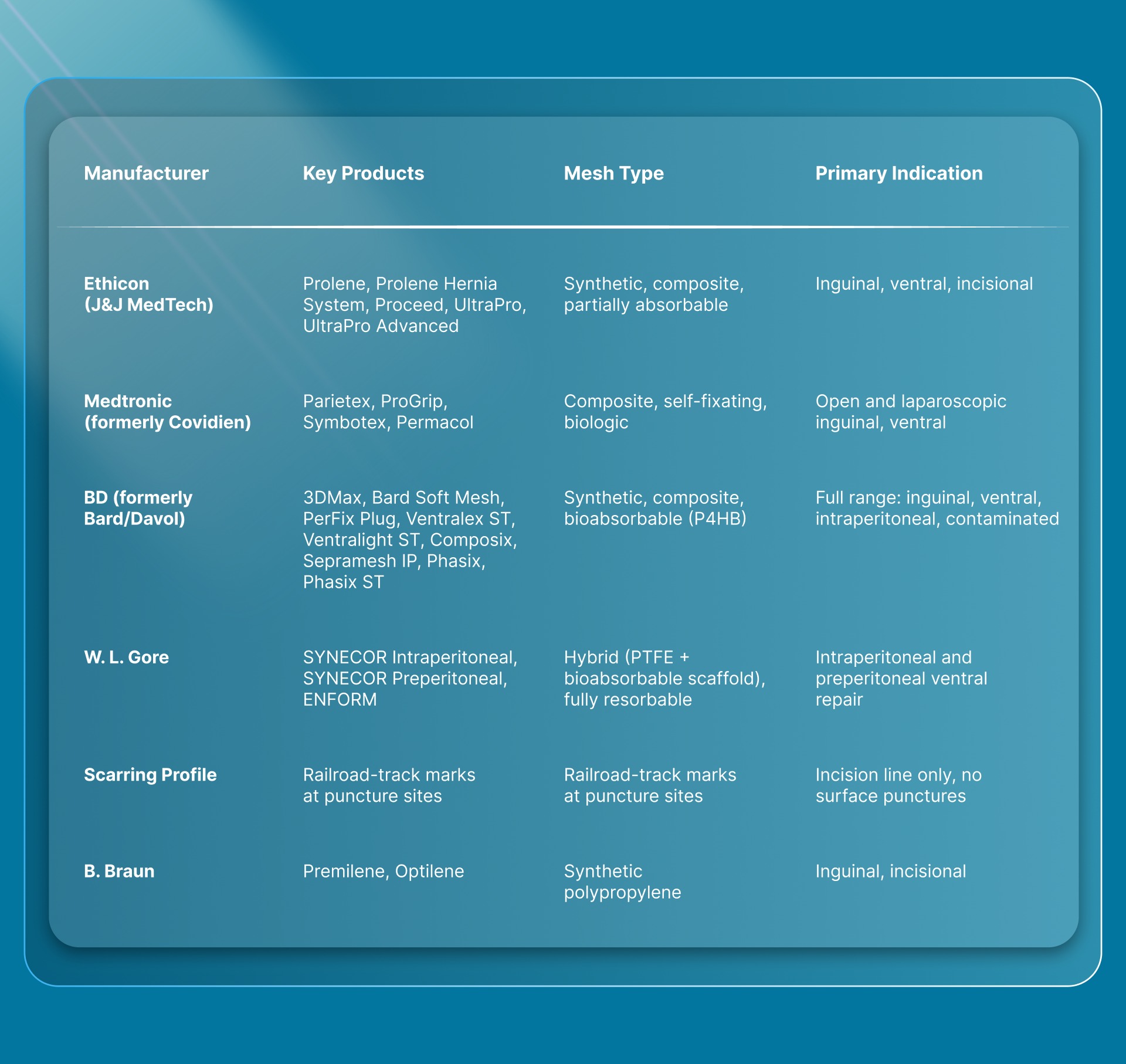
Here's what each manufacturer brings to hernia repair.
Ethicon (J&J MedTech)
Ethicon's hernia line includes polypropylene flat mesh (Prolene), a bilayer anterior-posterior system (Prolene Hernia System), composite mesh with an oxidized regenerated cellulose barrier (Proceed), and partially absorbable lightweight options (UltraPro, UltraPro Advanced). After partial absorption of its poliglecaprone-25 component, UltraPro leaves 65% less foreign material than microporous mesh.
The lineup covers inguinal, ventral, and incisional repairs. UltraPro is a common pick for surgeons who want reduced long-term foreign body presence.
Medtronic (Formerly Covidien)
Medtronic's portfolio includes Parietex (composite polyester), Symbotex (composite for laparoscopic repair), Permacol (biologic porcine dermis), and ProGrip, a self-fixating mesh with resorbable polylactic acid microgrips. ProGrip lets surgeons position and secure the mesh without tacks or sutures, and roughly 40% of the mesh weight resorbs over time.
ProGrip comes in both open and laparoscopic configurations. It's a popular choice for high-volume inguinal repair practices looking to cut fixation time.
Becton Dickinson (BD, Formerly Bard/Davol)
BD carries the broadest single portfolio in hernia mesh. Synthetic options include 3DMax, 3DMax Light, Bard Soft Mesh, PerFix Plug, and PerFix Light Plug. Composite products for intraperitoneal use include Composix L/P, Composix E/X, Sepramesh IP, Ventralex ST, and Ventralight ST.
The Phasix line is BD's bioabsorbable offering, made from poly-4-hydroxybutyrate (P4HB). The Phasix family has over 143,000 implants worldwide. Phasix ST adds a hydrogel barrier (Sepra technology) for intra-abdominal placement.
W. L. Gore
Gore focuses on hybrid biomaterial technology. SYNECOR Intraperitoneal Biomaterial uses a tri-layer design with a macroporous PTFE knit core and bioabsorbable layers on both surfaces. SYNECOR Preperitoneal Biomaterial uses the same PTFE knit with a bioabsorbable web for preperitoneal placement.
ENFORM Biomaterial is Gore's fully resorbable option. Gore's products target ventral hernia repairs where intraperitoneal or preperitoneal mesh placement is needed.
B. Braun
B. Braun offers Premilene (standard polypropylene) and Optilene (elastic monofilament polypropylene). The lineup is narrower than the other four manufacturers, focused on inguinal and incisional repair. B. Braun positions itself as a cost-effective polypropylene supplier with strong European clinical data.
How Can Surgeons Choose the Right Mesh for Each Hernia Type?
The right mesh depends on the hernia location, the contamination grade of the surgical field, and whether the repair is open or laparoscopic. No single brand or material covers every scenario.
Inguinal Hernia Repair
Lightweight synthetic polypropylene mesh is the standard for elective inguinal hernia repair in clean fields. Self-fixating meshes (with resorbable microgrips) reduce operative time by removing the need for separate tack or suture fixation during laparoscopic inguinal repair.
Synthetic mesh has driven inguinal hernia recurrence rates well below those of suture-only repair. The choice between flat mesh, plug-and-patch, and bilayer systems depends on the surgeon's preferred technique (Lichtenstein, TEP, or TAPP) and the patient's anatomy.
Ventral and Incisional Hernia Repair
Ventral and incisional hernias cover a wider range of defect sizes and contamination levels. In clean cases, synthetic or composite mesh placed in the sublay (retromuscular) position produces the lowest recurrence rates.
In the JAMA Surgery PRICE trial, 13% of 253 patients with contaminated ventral hernias had a recurrence within two years. Synthetic mesh drove the lower end of that figure, producing significantly fewer recurrences than biologic mesh. This finding has shifted practice toward synthetic materials even in clean-contaminated fields.
For intraperitoneal placement (where mesh contacts bowel), a barrier-coated composite product is required to prevent adhesion formation. Fascial closure technique matters here too, since the fascial repair provides the primary structural strength while the mesh reinforces it.
Contaminated or Infected Fields
Contaminated hernia repairs present the hardest material decision. Biologic mesh was traditionally favored here because of its theoretical advantage in infected environments. Recent evidence has challenged that default.
The PRICE trial data showed that synthetic mesh significantly reduced recurrence compared to biologic mesh, even in contaminated ventral hernia cases. Combined with the substantially lower cost of synthetic products, many surgeons now consider synthetic sublay mesh a reasonable first choice in clean-contaminated and some contaminated fields, reserving biologics for grossly infected cases or patients with prior mesh explantation.
How Does Mesh Choice Affect Long-Term Patient Outcomes?
Recurrence and chronic pain are the two outcomes that matter most when comparing hernia mesh materials. Both vary by mesh type, hernia location, and surgical technique.
Recurrence Rates
Across 758 ventral hernia patients pooled from four randomized trials (2025 data), biologic mesh recurred at 24.6% while synthetic mesh recurred at 9.7%. Biologic repairs also had a higher reoperation rate (16.9% vs. 10.4%), though that gap didn't reach statistical significance.
These figures come from ventral hernia repair data. In inguinal repair, synthetic mesh has pushed recurrence rates well below those seen with suture-only tissue repair. The gap between synthetic and biologic performance is smaller in clean inguinal cases than in the ventral data, since inguinal recurrence rates with synthetic mesh are already low.
Chronic Postoperative Pain
Chronic postoperative inguinal pain (CPIP) remains a concern across all mesh types. Globally, about 17.01% of patients report it after inguinal repair (pooled from 29,466 patients in a 2024 analysis).
The North American rate is lower at 6.04%, likely reflecting differences in surgical technique and follow-up definitions. Mesh weight, fixation method, and nerve handling during dissection all influence this outcome.
Lightweight meshes were designed to reduce chronic pain by leaving less foreign material in the inguinal canal. The clinical evidence on lightweight vs. heavyweight chronic pain differences is still being consolidated in ongoing systematic reviews. Surgeons choosing between mesh types should weigh chronic pain risk alongside recurrence data.
Total Episode Costs
Biologic ventral hernia repair costs an average of $80,420 per patient in the PRICE trial's 2024 cost analysis. Synthetic repair averaged $61,036. Higher prosthetic expense and longer OR time with biologic products accounted for the gap, not complication-related costs.
For private practices and ambulatory surgery centers managing per-case margins, that cost gap is significant. Mesh material isn't the only variable in total episode cost. Closure method, OR time efficiency, and follow-up visit burden all contribute to the final per-patient figure.
What Role Does Incision Closure Play After Hernia Mesh Placement?
Every hernia repair ends with an incision that needs to be closed, whether it's a 5 mm laparoscopic port site or a 20 cm open midline wound. The closure method affects scarring, infection risk, follow-up appointments, and OR time.
Trocar sites of 10 mm or larger are where 96% of port-site hernias form, particularly in the umbilical region. Meticulous fascial closure at these sites is essential.
The skin closure that follows also matters. Traditional metal staples require a removal visit at 7 to 10 days and leave railroad-track scars at each puncture site.

SubQ It! is a bioabsorbable skin closure system that places subcutaneous (under the skin) dermal fasteners, which dissolve naturally with no removal visit required. The SubQ It! SU-10 carries 10 fasteners for incisions up to 10 cm, covering laparoscopic trocar sites.
The SubQ It! SU-25 carries 25 fasteners for incisions up to 25 cm, handling open hernia repair wounds. It's the only product in its category that works on very small laparoscopic incisions where surface-applied devices can't operate.
Skin closure with an absorbable stapler took 16.2 minutes on average in a 4,311-patient mastectomy series. Hand-sewn suture took 36.5 minutes for the same step, with no difference in surgical site infection between the two groups.
A roughly 20-minute difference per case adds up fast in a high-volume hernia practice. Types of skin closure deserve the same procurement attention as mesh selection, especially in practices running tight OR schedules.
Hernia surgeons who close three or four cases per day with hand-sewn subcuticular suture can recover meaningful OR time by switching to a bioabsorbable stapling system. The time recovered either reduces daily overtime or creates capacity for additional cases, and removing the staple-removal visit cuts patient follow-up burden for both the practice and the patient.
How Can Private Practices Cut Total Hernia Repair Costs?
Mesh cost is only one line item in a hernia repair episode. OR time, closure time, follow-up visits, and complication management all contribute to the per-patient total. Private practices and ambulatory surgery centers have more control over these variables than hospital-based programs do.
Synthetic mesh costs substantially less per unit than biologic mesh, and the difference grows once you add the longer OR time biologic repairs typically require. For practices looking to lower per-case spend, choosing synthetic mesh in appropriate cases makes the biggest impact.
Closure time adds up too. A closure method that saves 15 to 20 minutes per case translates directly into recovered OR minutes. In a private setting where OR time determines daily case capacity, those minutes represent either reduced staff overtime or room for an additional patient. Surgical skin stapler pricing is a fraction of mesh cost, making the return on a faster closure method easy to calculate.
Additionally, the follow-up burden also matters. Traditional metal staples require a 7 to 10 day removal visit that takes staff time, occupies an exam room, and creates a scheduling touchpoint for the patient.
Bioabsorbable subcutaneous fasteners dissolve on their own, removing that visit from the workflow entirely. For a practice doing 10 to 15 hernia repairs per week, removing staple-removal appointments frees significant clinic capacity.
Final Thoughts
Hernia mesh brand selection is a clinical and financial decision that affects recurrence, chronic pain, total episode cost, and patient satisfaction. The right mesh depends on hernia type, contamination grade, and whether the repair is open or laparoscopic. Getting the mesh right is essential, and giving the same attention to incision closure completes the picture.
Evaluate mesh and closure together when reviewing your hernia repair formulary. Synthetic polypropylene covers most clean-field repairs at the lowest cost. Composite products handle intraperitoneal placement. Reserve biologics for grossly contaminated cases. Then look at how you're closing the skin, and whether your current method adds unnecessary time, scarring, or follow-up visits.
SubQ It! is a bioabsorbable wound closure system that places dissolving subcutaneous fasteners, closing incisions from small laparoscopic ports (SU-10, up to 10 cm) to open hernia repair wounds (SU-25, up to 25 cm) with no removal visit and no railroad-track scars. Contact us today to request a custom quote.
FAQs
1. How Often Do Hernia Mesh Manufacturers Update Their Product Lines?
Major manufacturers introduce new mesh products or update existing lines every few years. Surgeons should check manufacturer catalogs annually and confirm current availability with their sales representatives before placing standing orders.
2. Can Surgeons Mix Mesh Brands With Fixation Devices From a Separate Manufacturer?
Yes, surgeons commonly pair a mesh product from one manufacturer with tacks, sutures, or adhesive fixation from another. Compatibility depends on mesh pore size, thickness, and the fixation method's intended tissue depth.
3. Does Mesh Pore Size Affect Hernia Repair Outcomes?
Yes, macroporous meshes (pore size above 1 mm) allow better tissue ingrowth and resist infection more effectively than microporous alternatives. Pore size influences mesh flexibility, foreign body response, and long-term integration with native tissue.
4. Are Biologic Meshes Reimbursed at the Same Rate as Synthetic Products?
No, reimbursement rates vary by payer and contract. Biologic meshes typically carry higher acquisition costs that are not always offset by higher procedural reimbursement, creating a per-case margin gap for practices using them routinely.
5. What Sterilization and Storage Requirements Apply to Hernia Mesh?
Hernia mesh products arrive pre-sterilized and sealed. Storage conditions vary by manufacturer, typically requiring a dry environment at room temperature. Surgeons should verify expiration dates and packaging integrity before use, especially with older inventory.
6. Should Surgeons Use a Separate Mesh for Recurrent Hernias?
Yes, recurrent hernia repair often benefits from a mesh placed in a tissue plane the original repair did not use. A failed anterior inguinal repair, for example, may be revised with a posterior (preperitoneal) approach using a fresh mesh in a different plane.


