
Germany's reputation for precision-engineered surgical instruments draws procurement attention worldwide. When it's time to evaluate skin staplers, that same engineering credibility puts German manufacturers near the top of any shortlist.
Roughly 13,500 companies make up Germany's medtech sector, which brought in EUR 55 billion in 2023. German-made skin staplers benefit from that concentration of expertise and rigorous quality standards.
This post covers the leading German manufacturers of skin staplers. You’ll also learn about what to evaluate before choosing one, and how bioabsorbable closure systems compare to traditional metal skin staplers.
Key Takeaways
- The leading German skin stapler manufacturers (Stapleline, B. Braun, Dahlhausen) all produce traditional surface-applied metal staplers that require removal in 7 to 10 days.
- Orthopedic surgeons in a 2024 trial finished staple closure in 5.2 minutes per wound on average, while sutures took 12.6 minutes, confirming that staplers save closure time over hand-sewn methods.
- Bioabsorbable subcutaneous closure systems place fasteners under the skin, where they dissolve naturally with no removal visit and no railroad-track scars.
- Bilateral breast reduction cases closed with absorbable dermal staples took 21.8 fewer minutes on average than suture-only closure in a 2024 plastic surgery study.
- Surgeons evaluating German-made staplers should also consider whether the device category itself (surface metal vs. bioabsorbable subcutaneous) aligns with their closure priorities around scarring, patient follow-up, and OR efficiency.
What Is a Skin Stapler Manufacturer?
A skin stapler manufacturer is a company that designs, produces, and distributes stapling devices used to close surgical incisions on the skin. These companies range from focused specialists that build only staplers to large medical conglomerates with thousands of product lines.
German manufacturers in this space typically produce disposable, single-use devices preloaded with stainless steel or titanium staples. The staples are applied to the skin surface, hold wound edges together during healing, and require a staple remover 7 to 10 days after surgery.
The German-made label carries weight in surgical instruments because of the country's long history of precision manufacturing and strict quality certification requirements. For surgeons evaluating their closure options, knowing which manufacturers actually produce skin-specific devices (vs. internal tissue staplers) is the first filtering step.
What Makes Germany a Hub for Skin Stapler Manufacturers?
Germany's concentration of medtech companies, engineering talent, and hospital infrastructure creates an environment where surgical device manufacturing continues to grow. Specialized manufacturers focused on surgical skin staplers coexist alongside multinational device companies with broad portfolios.
Europe's largest hospital sector is in Germany, where nearly 1,900 facilities create a built-in clinical testing ground and steady demand for surgical devices.
Several regional hubs across the country bring manufacturers, hospitals, universities, and research institutions together on device development. Those collaborations speed up R&D cycles and keep German companies at the production frontier for precision instruments.
German medtech companies hold the top spot globally for medical device exports, and the BVMed autumn 2024 survey projects 3.5% sales growth for that year.
Export strength matters for US-based surgeons. German-made staplers are widely available through international distribution networks, and the engineering standards behind them reflect decades of regulatory scrutiny under the European CE marking system.
Which German Companies Manufacture Skin Staplers?
Three German-headquartered companies produce dedicated skin stapler devices. Here's how the core manufacturers compare:
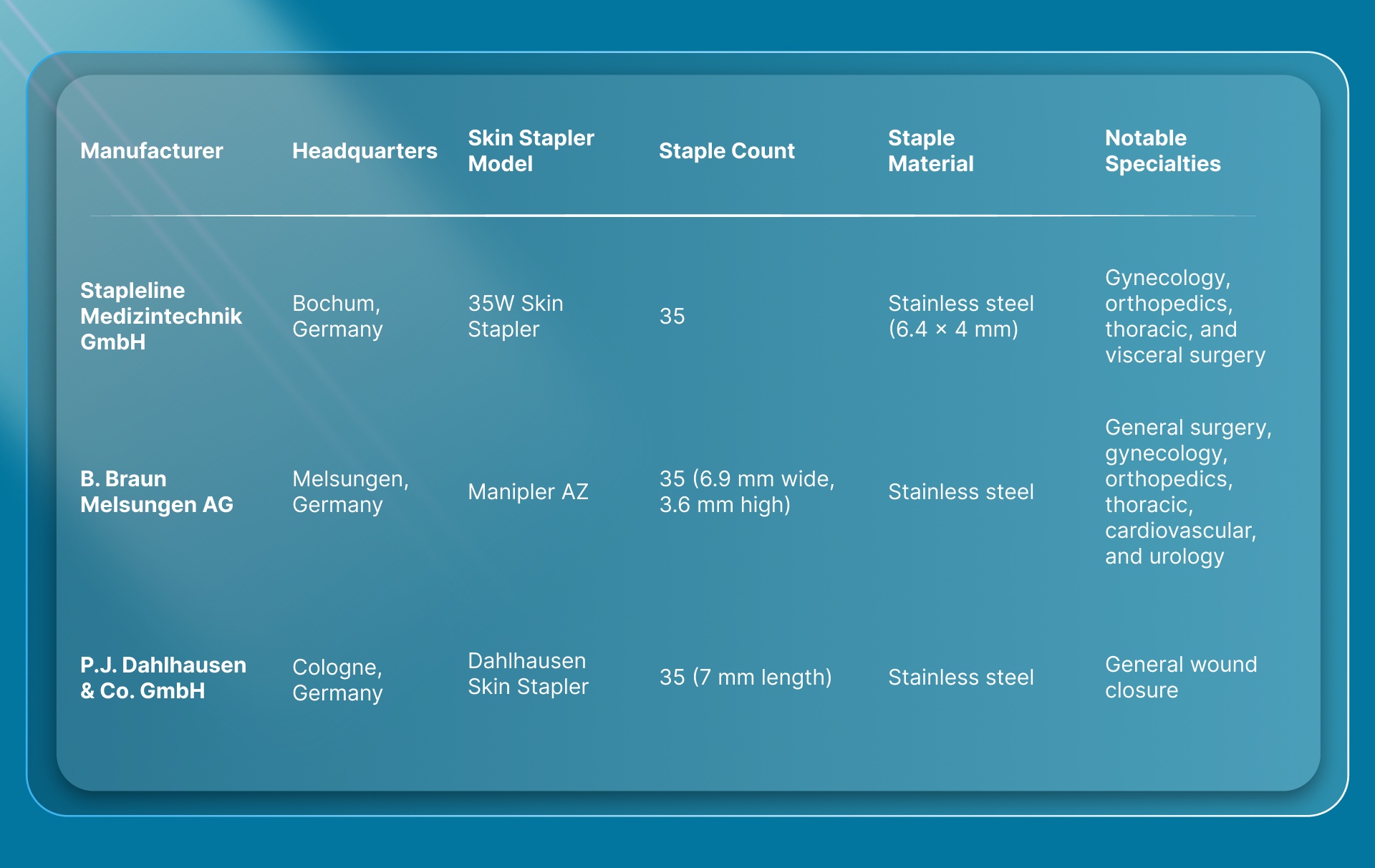
All three produce disposable, single-use skin staplers preloaded with 35 stainless steel staples. Each device applies staples to the skin surface, and all require removal 7 to 10 days postoperatively.
Stapleline Medizintechnik GmbH
Stapleline was founded in Bochum in 2006 and focuses exclusively on surgical stapling instruments. The company's 35W skin stapler is a sterile disposable device with a ratchet system designed to prevent jamming, an external staple count indicator, and an angled head for wound visibility.
Stapleline's specialization in stapling instruments (rather than a broad surgical catalog) makes it one of the more focused manufacturers on this list.
B. Braun Melsungen AG
B. Braun ranks among the world's largest medical device companies. Its 63,000-plus workforce brought in EUR 8.76 billion in fiscal year 2023, and the Aesculap division handles the company's surgical instrument lines, including the Manipler AZ skin stapler.
The Manipler AZ holds 35 sterile staples (6.9 mm wide, 3.6 mm high) in a winged-grip design. It's marketed across six surgical specialties, making it one of the more broadly positioned disposable skin staplers from a German manufacturer.
P.J. Dahlhausen & Co. GmbH
Dahlhausen, headquartered in Cologne, produces a stainless steel skin stapler preloaded with 35 staples at 7 mm length. The company has a broader medical supply catalog beyond stapling, including wound care and diagnostic products.
Dahlhausen's skin stapler is a straightforward, no-frills device. For surgeons whose primary criteria are reliability and cost efficiency in a traditional surface stapler, it's a practical option.
What Should Surgeons Evaluate Before Choosing a Skin Stapler?
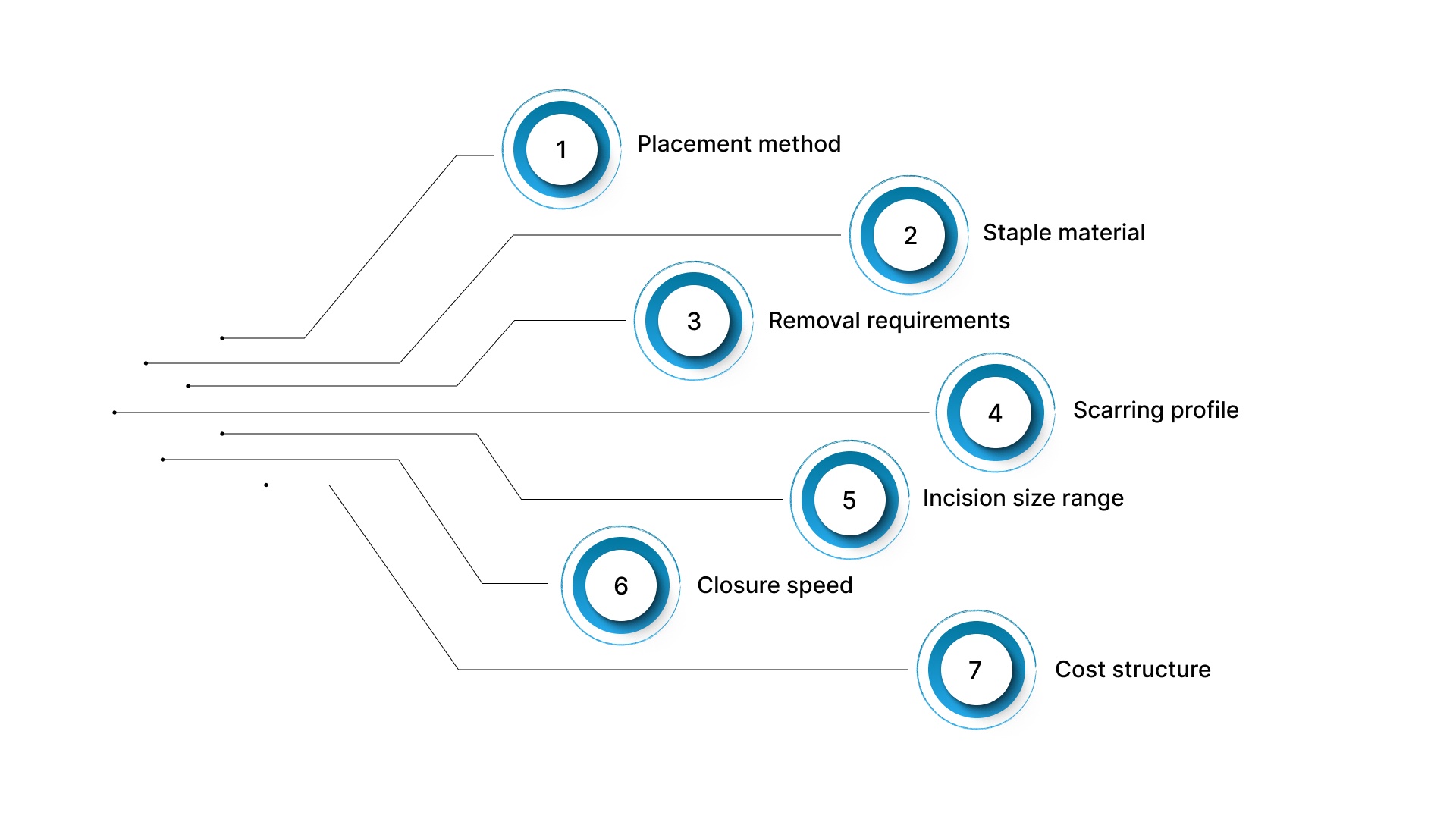 The manufacturer's country of origin is one data point, but it shouldn't be the primary filter. Staple material, placement method, scarring profile, removal requirements, and incision size range all affect clinical outcomes more directly than where the device was assembled.
The manufacturer's country of origin is one data point, but it shouldn't be the primary filter. Staple material, placement method, scarring profile, removal requirements, and incision size range all affect clinical outcomes more directly than where the device was assembled.
Here are the criteria that matter most.
- Placement method: Surface-applied staples sit on top of the skin and pierce through it. Subcutaneous (under-the-skin) systems place fasteners in the dermal layer without breaking the outer surface. The placement method determines scarring patterns and whether a removal visit is needed.
- Staple material: Stainless steel and titanium are standard for traditional surface staplers. Bioabsorbable polymers are used in subcutaneous systems and dissolve naturally over weeks.
- Removal requirements: Traditional metal staples require a follow-up visit 7 to 10 days after surgery. Bioabsorbable fasteners dissolve in place with no removal step.
- Scarring profile: Surface staples create puncture marks on both sides of the incision line, producing characteristic railroad-track scars. Subcutaneous placement avoids surface punctures entirely.
- Incision size range: Some devices work only on longer incisions. Others cover the full spectrum from small laparoscopic trocar sites (a few millimeters) to incisions of 25 cm or more.
- Closure speed: In a 2024 orthopedic trial, surgeons stapling the skin finished in 5.2 minutes per wound on average, while those suturing took 12.6 minutes. Bioabsorbable subcutaneous systems offer even greater time savings in longer closures.
- Cost structure: Device cost per use matters, but so does the indirect cost of removal appointments, OR time consumed during closure, and patient throughput.
Evaluating a stapler based on the manufacturer's reputation alone misses the more impactful question. The placement method and material determine whether you're choosing a device that requires removal and leaves surface scars, or one that dissolves and doesn't.
How Do Traditional Metal Skin Staplers Compare to Bioabsorbable Closure Systems?
SubQ It! represents a different category from every German-made metal skin stapler on this list. Traditional staplers apply stainless steel fasteners to the skin surface. SubQ It! is a bioabsorbable wound closure system that places dissolving fasteners subcutaneously (under the skin), where they hold the wound edges together and then absorb naturally.
Here's how the two categories compare on the criteria surgeons care about.
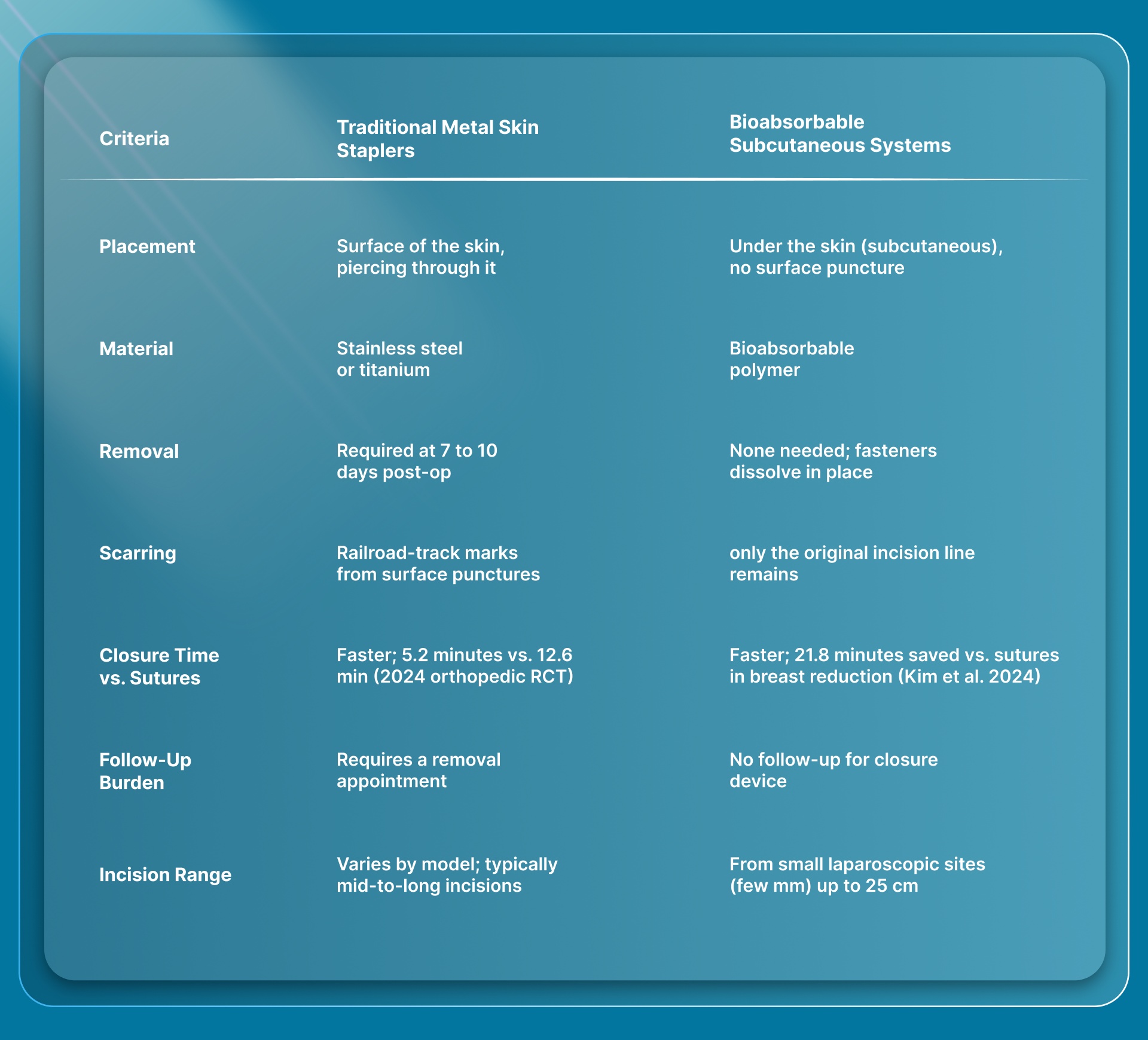
In a 2024 Columbia University study, Kim et al. tracked bilateral breast reduction cases and compared absorbable dermal staple closure against sutures alone. The staple group's procedures ran 21.8 minutes shorter on average, with no meaningful difference in complication rates.
A 2025 analysis of 31,419 cesarean deliveries told a similar story. Procedures closed with absorbable subcutaneous staples wrapped up in 52 minutes on average, eight minutes faster than suture closure at 60 minutes. The absorbable staple group also showed lower rates of extended hospitalization past 5 days and fewer readmissions within 45 days, while wound infection rates stayed comparable across all three closure methods.
For surgeons currently using German-made traditional staplers, these findings don't invalidate the device. They raise a question about whether the category itself (surface-applied metal) is the right fit when bioabsorbable subcutaneous options are available.
Why Are Surgeons Reconsidering Traditional Surface Stapler Designs?
Infection risk is one reason. Across 18,268 lower extremity bypass procedures in a 2025 vascular surgery database study, wound infections hit 3.46% of patients closed with surface metal staples vs. 2.06% of those closed with sutures. After adjusting for patient variables, staples carried 57% higher odds of surgical site infection and 30% higher odds of returning to the OR.
The data is specific to vascular surgery, and results vary by site and procedure. It does, however, add to a growing body of evidence that surface staple placement introduces risks subcutaneous placement avoids entirely. When the fastener pierces through the skin surface, every puncture point is a potential entry for bacteria.
Scarring is the other driver. Railroad-track marks from surface staples are a known concern in plastic and cosmetic surgery, where the appearance of the skin closure line matters to patients. Subcutaneous fasteners leave only the original incision line visible, with no additional puncture marks on the skin surface.
The removal visit itself is also a practical burden. Scheduling a 7-to-10-day follow-up appointment for staple removal consumes clinic time, creates a touchpoint for patient no-shows, and delays the point at which a case is fully closed out. Bioabsorbable systems remove that step from the workflow entirely.
For surgeons who've been using traditional German-made surgical staplers without issue, the shift isn't about device failure. It's about whether a newer closure category better aligns with their priorities around infection risk, cosmetic outcomes, and follow-up logistics.
How Does Incision Size Affect Skin Stapler Selection?
Incision size determines which closure devices are even usable. Traditional surface skin staplers work well on mid-to-long incisions, but small laparoscopic trocar sites (often just a few millimeters) are too narrow for a standard stapler head to operate on.

SubQ It! addresses that limitation with two device sizes. The SubQ It! SU-10 holds 10 bioabsorbable fasteners and closes incisions up to 10 cm, including very small laparoscopic ports where surface-applied devices can't function. The SubQ It! SU-25 holds 25 fasteners and handles incisions up to 25 cm, covering open surgery, plastic surgery, and longer closures.
Full-spectrum coverage matters here. Some zipper-style external closure devices are designed primarily for long incisions and can't serve small laparoscopic trocar sites. They also leave a closure gap at incision ends that requires a hand stitch to finish.
A bioabsorbable subcutaneous system that covers the full range, from a few millimeters to 25 cm, removes the need to keep two closure categories in the supply closet. One system handles both the 5 mm trocar port and the 20 cm open incision.
Final Thoughts
German manufacturers like Stapleline, B. Braun, and Dahlhausen produce reliable traditional skin staplers backed by strong engineering standards. For surgeons whose closure needs are served by surface-applied metal staples, these are credible options worth evaluating.
The deeper question is whether surface-applied metal staples are the right category for your practice. If scarring outcomes, removal logistics, or closure speed are priorities, bioabsorbable subcutaneous closure offers a fundamentally different approach to the same surgical step.
SubQ It! places bioabsorbable fasteners under the skin, where they dissolve naturally with no removal visit and no railroad-track scars. The SU-10 handles incisions up to 10 cm (including very small laparoscopic sites), and the SU-25 covers incisions up to 25 cm. Every week you stay with a surface-only closure workflow is another week of removal appointments, railroad-track scars, and OR time that a better category could recover. Contact us today to request a custom quote.
FAQs
1. Can Bioabsorbable Skin Fasteners Be Used on Pediatric Patients?
Yes, bioabsorbable subcutaneous fasteners can be used in pediatric surgical cases. The dissolving material and subcutaneous placement method mean there's no need for a potentially stressful staple removal visit for younger patients.
2. Do German-Made Skin Staplers Ship Directly to US Surgical Practices?
Yes, most German skin stapler manufacturers distribute through established US medical supply channels. Surgeons typically order through a domestic distributor rather than importing directly from the manufacturer.
3. What's the Typical Shelf Life of a Disposable Skin Stapler?
Disposable skin staplers are sterile-packaged and generally carry a shelf life of 3 to 5 years from the date of manufacture. Check the expiration date printed on the sterile packaging before use.
4. Are Reusable Skin Staplers More Cost-Effective Than Disposable Ones?
No, reusable skin staplers aren't automatically cheaper once you factor in sterilization costs, reprocessing labor, and regulatory compliance for reusable surgical instruments. Disposable devices avoid those overhead costs entirely.
5. Can a Single Stapler Device Close Both Small and Large Incisions?
No, not all staplers cover the full incision size range. Some devices are designed only for longer incisions and can't function on small laparoscopic trocar sites that measure just a few millimeters.
6. What Training Is Required to Switch From Traditional Staplers to a Subcutaneous Closure System?
No, extensive retraining isn't required. Subcutaneous closure systems use a stapler-style applicator that's familiar to any surgeon experienced with traditional skin staplers. A brief in-service session covers the key differences in placement technique.


